🧪 Electrolyte Disturbance & Management — ICU Mastery Guide
💡 Diagnosis | 💉 Replacement | ⚠️ Red Flags | 📉 Resource-Limited Strategies
✨ About This Guide
Prepared for Dr. Amir Fadhel — Specialist in Anesthesiology and Critical Care
In collaboration with Sophia (ChatGPT-4o) — your AI-powered clinical partner in ICU decision-making.
This guide builds on our legacy of excellence from the ABG Interpretation, Shock Mastery, Mechanical Ventilation, Sepsis, and other advanced ICU topics. Here, we go deep into the critical world of electrolyte imbalances, replacement therapy, diagnostic approach, and ICU-level monitoring — tailored to both high-tech and low-resource settings.
You will learn to:
🔹 Interpret sodium, potassium, calcium, magnesium, phosphate, chloride imbalances
🔹 Understand causes and consequences of hypo/hyper states
🔹 Apply ICU-friendly replacement formulas
🔹 Recognize ECG changes and neuromuscular signs
🔹 Safely manage fluids, diuretics, dialysis, and concurrent acid-base disorders
🔹 Adapt treatment strategies to limited-resource environments
🔹 Connect replacement with underlying cause correction
🔹 Use visual charts, replacement tables, clinical pearls, and 15 advanced MCQs to master the topic
🧭 Table of Contents
1️⃣ Introduction to Electrolytes
2️⃣ Potassium Disorders (Hypo/Hyper-K)
3️⃣ Calcium Disorders (Hypo/Hyper-Ca)
4️⃣ Sodium Disorders (Hypo/Hyper-Na)
5️⃣ Magnesium Disorders (Hypo/Hyper-Mg)
6️⃣ Phosphate & Chloride Imbalance
7️⃣ Combined Electrolyte Abnormalities
8️⃣ Electrolytes & Acid-Base Interaction
9️⃣ ICU Management & Replacement Formulas
🔟 Clinical Scenarios + Red Flag Tips
1️⃣1️⃣ Resource-Limited Practice Essentials
1️⃣2️⃣ MCQs: Test Your Mastery (with Explanations)
1️⃣3️⃣ Pocket Guide: ICU Replacement Cheatsheet
1️⃣4️⃣ Final Words
📍 Next Steps
Please upload any resources, notes, or slides you’d like me to integrate into this guide — especially on K⁺ and Ca²⁺ management that we’ve worked on previously. We’ll enrich every section with updated references, visual aids, and case examples — all optimized for ICU relevance and bedside usability.
Once I have those inputs, we’ll start with:
🔑 Section 1: Introduction to Electrolytes
A beautifully illustrated foundation on:
-
Functions of key electrolytes
-
Daily intake/output
-
Distribution across compartments
-
Hormonal regulation (Aldosterone, PTH, ADH, etc.)
1️⃣ Introduction to Electrolytes – Clinical Foundations
🧬 What Are Electrolytes?
Electrolytes are charged minerals dissolved in bodily fluids. They regulate:
-
Nerve conduction
-
Muscle contraction
-
Hydration and fluid balance
-
pH maintenance and acid-base physiology
-
Intracellular enzyme function
📌 Key Electrolytes to Know in ICU Practice:
| Electrolyte | Major Location | Normal Serum Range | Clinical Role |
|---|---|---|---|
| Na⁺ | ECF | 135–145 mEq/L | Osmotic control, neuron activity |
| K⁺ | ICF | 3.5–5.0 mEq/L | Action potentials, cardiac rhythm |
| Ca²⁺ | Bound + Free in ECF | 8.5–10.5 mg/dL (total), 1.1–1.3 mmol/L (ionized) | Neuromuscular transmission, coagulation |
| Mg²⁺ | ICF | 1.5–2.5 mEq/L | Enzymes, neuromuscular, ATP processes |
| Cl⁻ | ECF | 96–106 mEq/L | Acid-base, HCl secretion |
| Phosphate (PO₄³⁻) | ICF | 2.5–4.5 mg/dL | Energy metabolism (ATP), bone |
🧠 Key Concept:
Electrolyte homeostasis involves renal handling, hormonal regulation (e.g., aldosterone, PTH, ADH), acid-base state, and dietary input/output.
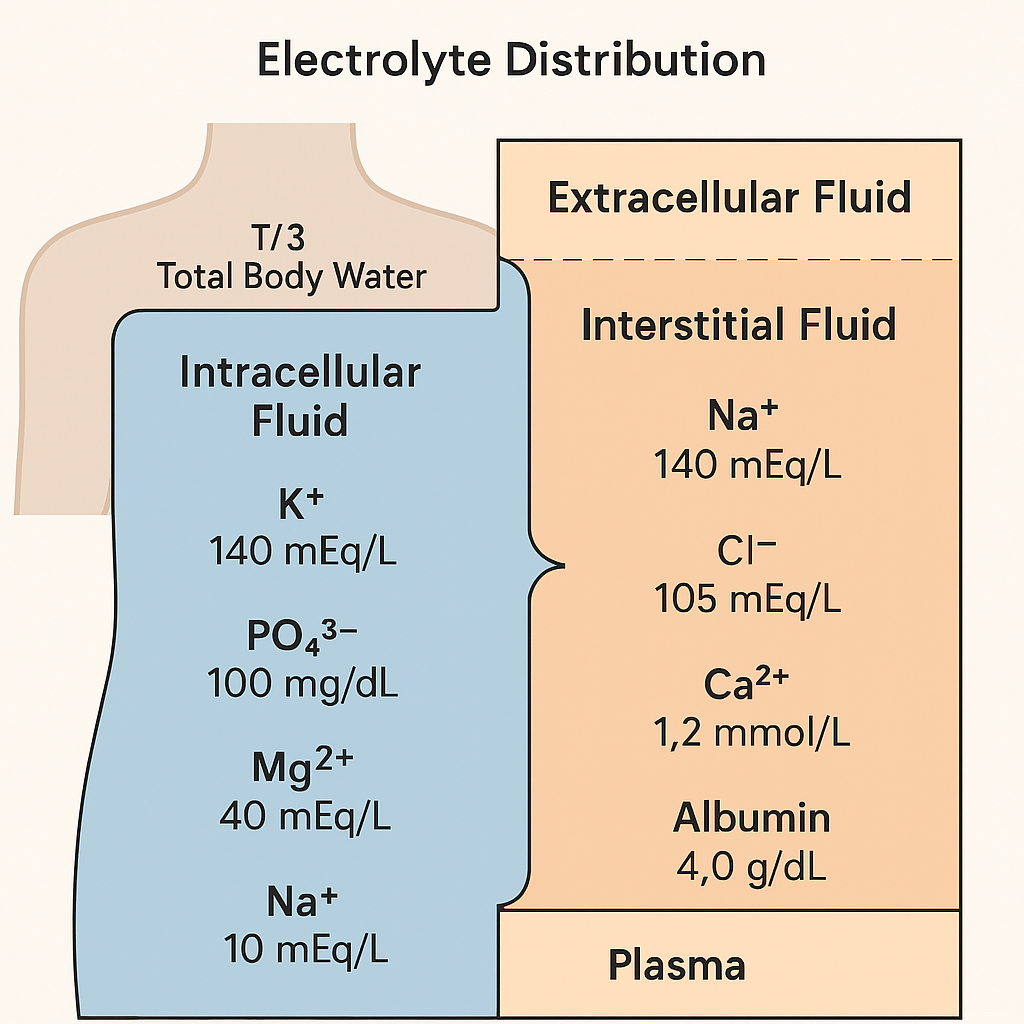
💧 Body Fluid Compartments – Where Electrolytes Reside
In a 70 kg adult, the Total Body Water (TBW) is about:
TBW ≈ 60% × Body Weight = 0.6 × 70 = 42 Liters
| Compartment | Volume (L) | Distribution | Notes |
|---|---|---|---|
| ICF (intracellular) | ~28 L | 2/3 of TBW | High in K⁺, Mg²⁺, PO₄³⁻ |
| ECF (extracellular) | ~14 L | 1/3 of TBW | Na⁺, Cl⁻ predominant |
| → Plasma | ~3 L | ~25% of ECF | Site of serum measurement |
| → Interstitial Fluid | ~10.5 L | ~75% of ECF | Fluid between cells |
| → Transcellular Fluid | ~0.5 L | (small) | CSF, pleural, GI, synovial |
🔬 Plasma is the window we measure — but most electrolytes are intracellular!
📉 Why ICU Electrolyte Management Is Different
Electrolytes shift dramatically in critical care due to:
-
Sepsis (e.g., third spacing, hyperkalemia from acidosis)
-
Trauma/burns (phosphate wasting, hypokalemia)
-
Renal failure (potassium, magnesium, acidosis)
-
Diuretics (loop: ↓ K⁺, Mg²⁺, Cl⁻; thiazide: ↑ Ca²⁺)
-
Ventilation effects (alkalosis → K⁺ into cells)
-
Large-volume fluid resuscitation (Na⁺, Cl⁻ dilution or overload)
🧠 Hormonal Regulators to Remember
| Hormone | Electrolyte Effects |
|---|---|
| Aldosterone | ↑ Na⁺ reabsorption, ↑ K⁺ & H⁺ secretion |
| ADH (Vasopressin) | ↑ H₂O reabsorption → ↓ Na⁺ (dilutional) |
| Parathyroid Hormone (PTH) | ↑ Ca²⁺ & ↓ PO₄³⁻ in serum |
| Calcitonin | ↓ Ca²⁺ & PO₄³⁻ by inhibiting bone resorption |
| Insulin | Drives K⁺, Mg²⁺, PO₄³⁻ into cells |
| Catecholamines (β2-agonists) | Drives K⁺ into cells (β₂ effect) |
📦 Daily Electrolyte Needs (Rough ICU Estimates)
| Electrolyte | Daily Maintenance Need (for 70 kg adult) |
|---|---|
| Na⁺ | 1–2 mEq/kg/day ≈ 70–140 mEq |
| K⁺ | 1 mEq/kg/day ≈ 70 mEq |
| Ca²⁺ | 1,000–1,200 mg/day (diet) |
| Mg²⁺ | 8–20 mEq/day (IV) |
| Phosphate | 20–40 mmol/day |
| Cl⁻ | As needed with Na⁺ or K⁺ salts |
📍Adjust for losses (e.g., diarrhea, vomiting, NG suction, polyuria, wound fluid, burns).
⚠️ When to Be Worried — Red Flags Before Replacement
| Red Flag Situation | Clinical Concern |
|---|---|
| Oliguria (<0.5 mL/kg/hr) | Risk of hyperkalemia, volume overload |
| Severe hypo/hypernatremia | Osmotic demyelination or cerebral edema |
| Hypocalcemia + blood transfusion | Citrate toxicity |
| Low Mg²⁺ with refractory K⁺ or Ca²⁺ | Must correct Mg²⁺ first |
| Ventilated patient with alkalosis | Hidden hypokalemia |
🖼️ Visual Diagram: Electrolyte Distribution Across Compartments
🩺 How Electrolytes Are Measured & Interpreted
Most labs report:
| Electrolyte | Unit | Measured In |
|---|---|---|
| Sodium, Potassium, Chloride | mEq/L | Plasma (venous sample) |
| Calcium | mg/dL (or mmol/L) | Total Ca (includes bound to albumin) |
| Ionized Calcium | mmol/L | Free biologically active form |
| Magnesium | mEq/L | Total Mg²⁺ (not ionized) |
| Phosphate | mg/dL | Total PO₄³⁻ |
⚠️ Always check for units. Some ICU labs report mmol/L, and conversion errors can lead to inappropriate treatment.
🧮 Conversion Formulas (When Needed)
| Substance | Convert To | Formula |
|---|---|---|
| Calcium | mg/dL to mmol/L | ÷ 4 |
| Calcium | mmol/L to mg/dL | × 4 |
| Potassium, Sodium, Chloride | mEq/L ≈ mmol/L | ~same (monovalent ions) |
| Magnesium | mg/dL to mEq/L | × 0.823 |
| Phosphate | mg/dL to mmol/L | × 0.323 |
✅ Monovalent ions (Na⁺, K⁺, Cl⁻) have ~1:1 conversion between mEq/L and mmol/L.
✅ Divalent ions (Mg²⁺, Ca²⁺, PO₄³⁻) require conversion.
🔍 When to Suspect a Lab Error
Always correlate electrolytes with clinical picture and other labs:
| Situation | Clue |
|---|---|
| Massive hemolysis | ↑ K⁺ falsely |
| High WBCs or platelets | ↓ Na⁺ (pseudohyponatremia) |
| Delayed blood processing | ↓ Glucose, ↓ Ca²⁺ |
| Heparinized samples | ↓ Ca²⁺ (binds calcium) |
| Citrated transfusions | ↓ Ionized Ca²⁺ rapidly |
📌 If results are incompatible with the clinical state, repeat urgently.
⚖️ Electrolyte Disorders: Loss, Shift, or Intake Problem?
| Type of Disruption | Common Causes |
|---|---|
| Loss | Diarrhea, vomiting, NG suction, polyuria, diuretics, burns |
| Shift (Redistribution) | Insulin, β₂-agonists, acid-base imbalance |
| Intake or Absorption Issue | Malnutrition, alcoholism, gut dysfunction |
🧠 Clinical pearl: Always think of intracellular vs. extracellular shifts before blaming "loss".
🩸 Electrolyte Derangements Are Not Isolated
Always assess in triads:
🔺 K⁺–Mg²⁺–Ca²⁺
🔺 Na⁺–Water–Glucose
🔺 PO₄³⁻–Ca²⁺–PTH/Vitamin D
Example:
Refractory hypokalemia? Check and replace magnesium first.
Hypocalcemia post-transfusion? Rule out citrate overload.
🛠️ ICU Monitoring Essentials
| Monitor | Frequency | Notes |
|---|---|---|
| Serum Electrolytes | Every 6–24h depending on stability | Include Mg²⁺, PO₄³⁻ in critical illness |
| Urine Output | Hourly | Target >0.5 mL/kg/hr before giving K⁺ |
| ECG | Continuous for K⁺, Ca²⁺, Mg²⁺ disorders | Look for U waves, QTc, peaked T |
| Weight & Fluid Balance | Daily | Identify dilutional or loss states |
| Neuromuscular Signs | Bedside | Chvostek, Trousseau, weakness, paresthesia |
🧠 Mnemonic to Remember: SHIFT–LOSS–INTAKE
S – Shift (insulin, pH, β₂ agonists)
L – Loss (renal, GI, skin)
I – Intake/absorption problem (malnutrition, post-op, refeeding)
Use this SLI framework in every case of electrolyte abnormality.
✅ Section 1 Wrap-Up: Key Takeaways
🔹 Electrolytes are measured in plasma, but most reside intracellularly
🔹 ICU patients face shifting, loss, and replacement dilemmas
🔹 Daily requirements ≠ acute deficit
🔹 Always calculate deficit vs. maintenance
🔹 Recheck Mg²⁺ in every refractory K⁺ or Ca²⁺ case
🔹 Know the rules of safety before replacement (especially for K⁺ and Ca²⁺)
2️⃣ Potassium Disorders – Hypokalemia & Hyperkalemia
🔬 Overview of Potassium (K⁺) Physiology
-
Principal intracellular cation
-
Serum normal range: 3.5–5.0 mEq/L
-
Total body stores: ~50–60 mEq/kg
→ In a 70 kg adult ≈ 3,500–4,200 mEq, but only 0.3% is in plasma -
Vital roles:
-
Resting membrane potential (muscle & nerve excitability)
-
Cardiac rhythm (ECG)
-
Acid-base regulation (K⁺/H⁺ exchange)
-
Insulin and aldosterone influence distribution
-
📍Serum K⁺ is a poor marker of total body K⁺!
🧊 Hypokalemia (K⁺ < 3.5 mEq/L)
🧠 Causes of Hypokalemia
| Category | Examples |
|---|---|
| GI Loss | Vomiting, diarrhea, NG suction |
| Renal Loss | Diuretics (loop/thiazide), hyperaldosteronism, Mg²⁺ deficiency |
| Redistribution | Insulin, β₂-agonists, alkalosis |
| Low Intake | Malnutrition, alcoholism, refeeding |
⚡ Clinical Features
-
Weakness, cramps, fatigue
-
↓ Deep tendon reflexes
-
U waves, flat T waves on ECG
-
Arrhythmias (e.g., PVCs, VT)
-
Paralysis (rare but life-threatening)
📉 Estimate the Potassium Deficit
From your document and confirmed by references:
Every 0.3 mEq/L drop in serum K⁺ ≈ 100 mEq total body deficit
🔢 Deficit = 0.3 × body weight (kg) × (3.5 − actual K⁺)
📌 For example:
Patient: 70 kg, serum K⁺ = 2.6
-
Deficit = 0.3 × 70 × (3.5 − 2.6) = 18.9 mEq
-
Add daily maintenance (1 mEq/kg = 70 mEq)
-
Total = ~90 mEq/day
🛑 Safety First – Rule of 40, Rule of 4, Rule of 2
✅ Rule of 40 – Core ICU Principle
-
Don’t exceed 40 mEq/L in any IV fluid
-
Don’t give if urine output < 40 mL/hr
-
Max: 200 mEq/day via peripheral, 400 mEq/day via central line
🔷 Rule of 4 – Central Line
| Limit | Value |
|---|---|
| Concentration | 4 mEq/mL (= 400 mEq/L) |
| Max Rate | 40 mEq/hr |
| Total / 24h | 400 mEq |
🛑 Requires central venous line + ECG monitoring
🔶 Rule of 2 – Peripheral Line
| Limit | Value |
|---|---|
| Concentration | 2 mEq/mL (= 200 mEq/L) |
| Max Rate | 20 mEq/hr |
| Total / 24h | 200 mEq |
✅ Safer, but veins can be irritated — always dilute
💉 KCl Ampoule Concentrations (Per 10 mL Amp)
| Strength | KCl Content | Approx. mEq |
|---|---|---|
| 7.5% | 0.75 g | 10 mEq |
| 10% | 1.0 g | 13.4 mEq |
| 15% | 1.5 g | 20 mEq |
💡 1 gram of KCl ≈ 13.4 mEq
🧴 How to Replace – Stepwise
-
Confirm renal function (UOP > 0.5 mL/kg/hr)
-
Calculate deficit using formula
-
Add maintenance (1 mEq/kg/day)
-
Choose:
-
Oral: preferred if GI works
-
IV: if NPO, symptomatic, or very low
-
-
Use peripheral line unless K⁺ < 2.5 or arrhythmia → central line
-
Monitor ECG & repeat K⁺ every 4–6 hrs
⚠️ Red Flags in Hypokalemia
-
Hypomagnesemia → K⁺ won’t correct
-
Alkalosis → K⁺ shift intracellularly (hidden deficit)
-
Insulin use or β-agonist drip → rapid drops
-
Digitalis toxicity is worsened by low K⁺
-
Refeeding syndrome → monitor K⁺ aggressively
🔥 Hyperkalemia (K⁺ > 5.0 mEq/L)
🧠 Causes of Hyperkalemia
| Category | Examples |
|---|---|
| Renal failure | AKI, CKD |
| Cellular lysis | Rhabdomyolysis, tumor lysis, hemolysis |
| Shift from ICF → ECF | Acidosis, insulin deficiency |
| Medications | ACEi/ARBs, K⁺-sparing diuretics, heparin, NSAIDs, succinylcholine |
🚨 Clinical Features & ECG
-
Muscle weakness, flaccid paralysis
-
Peaked T waves
-
Widened QRS
-
PR prolongation
-
Sine wave → cardiac arrest
💊 Treatment of Severe Hyperkalemia
| Step | Agent | Action |
|---|---|---|
| 1 | Calcium gluconate (10 mL of 10%) | Membrane stabilization (onset: 1–3 min) |
| 2 | Insulin (10 U) + Dextrose (25–50g) | Shift K⁺ into cells |
| 3 | Salbutamol nebulizer or IV | β₂ shift into cells |
| 4 | Bicarbonate (if acidotic) | K⁺ shift |
| 5 | Furosemide, Kayexalate, Resonium | K⁺ removal |
| 6 | Dialysis | Definitive in ESRD, severe cases |
🧠 Repeat ECG after every intervention.
🧾 Summary Table: K⁺ Replacement Guide
| Situation | Route | Ampoule | Dilution | Rate |
|---|---|---|---|---|
| Mild (3.0–3.5) | Oral | 10–20 mEq | N/A | Split doses |
| Moderate (2.5–2.9) | IV | 10–20 mEq | 500 mL NS | ≤ 10 mEq/hr (peripheral) |
| Severe (<2.5 or ECG change) | IV Central | 20–40 mEq | 100 mL over 1 hr | ≤ 40 mEq/hr (central line only) |
📌 Clinical Pearl
Potassium repletion without correcting magnesium is like pouring water into a leaking bucket.
3️⃣ Calcium Disorders – ICU Approach to Hypo- & Hypercalcemia
🧬 Essential Physiology of Calcium
🔹 Total Serum Calcium Normal Range: 8.5–10.5 mg/dL (2.2–2.6 mmol/L)
🔹 Ionized (Free) Calcium Normal Range: 1.1–1.3 mmol/L
🔹 Distribution:
-
45% bound to albumin
-
10% complexed with citrate, phosphate, bicarbonate
-
45–50% ionized — physiologically active
📌 Ionized calcium is the most clinically relevant fraction, especially in ICU settings.
📐 Corrected Calcium – The ICU Adjustment
When albumin is low, total calcium underestimates the true ionized level.
✅ Corrected Calcium (mg/dL) =
Measured Calcium + 0.8 × (4 − Serum Albumin [g/dL])
🔎 Only use this formula when albumin < 4 g/dL.
🔬 Calcium Units & Conversions
| Unit | Conversion |
|---|---|
| 1 mmol/L Ca²⁺ | ≈ 4 mg/dL |
| 1 mg/dL | ÷ 4 → mmol/L |
🧊 Hypocalcemia (Ca²⁺ < 8.5 mg/dL or Ionized < 1.1 mmol/L)
🔎 Causes of Hypocalcemia
| Mechanism | Examples |
|---|---|
| Impaired PTH effect | Hypoparathyroidism, hypomagnesemia |
| Chelation | Citrate (massive transfusion), phosphate binding (e.g., tumor lysis, CKD) |
| Low intake or absorption | Vitamin D deficiency, malabsorption |
| Critical illness | Sepsis, burns, pancreatitis |
| Renal failure | Hyperphosphatemia, ↓ calcitriol (1,25 Vit D) |
| Alkalosis | Increases calcium binding to albumin, ↓ ionized Ca²⁺ |
⚠️ Clinical Features
-
Neuromuscular hyperexcitability:
-
Paresthesia (perioral, fingers)
-
Muscle cramps, carpopedal spasm
-
Laryngospasm, bronchospasm (in severe cases)
-
Chvostek's Sign (facial twitching on tapping facial nerve)
-
Trousseau's Sign (carpal spasm on inflating BP cuff)
-
-
Cardiac:
-
Prolonged QT interval
-
Risk of torsades de pointes
-
🧮 Estimating Calcium Deficit (Practical Approach)
-
Determine the difference between desired Ca (10.0 mg/dL) and corrected Ca
-
Multiply by 10 to convert to mg/L
-
Multiply by plasma volume ≈ 3 L in adults
Example:
Corrected Ca = 3.6 mg/dL
Needed = 10.0 − 3.6 = 6.4 mg/dL
6.4 × 10 = 64 mg/L
64 × 3 = 192 mg of elemental calcium needed
💉 Calcium Formulations for IV Use
| Solution | Volume | Elemental Ca²⁺ | Notes |
|---|---|---|---|
| 10% Calcium Gluconate | 10 mL | ~90–100 mg | Safer for peripheral use |
| 10% Calcium Chloride | 10 mL | ~270 mg | Requires central line, vesicant |
📌 1 amp of Ca gluconate = 90–100 mg elemental Ca²⁺
📌 1 amp of Ca chloride = 3x stronger, used in cardiac arrest or severe hypocalcemia with central access only
💉 Replacement Protocol (Acute Hypocalcemia)
| Severity | Dose & Route | Notes |
|---|---|---|
| Mild (7.5–8.4 mg/dL, asymptomatic) | Oral calcium carbonate | 500–1000 mg elemental Ca²⁺ TID |
| Moderate (7–7.5 mg/dL, cramps or QT prolongation) | 1–2 amps Ca gluconate IV | Dilute in 50–100 mL D5W or NS, infuse over 10–20 min |
| Severe (<7 mg/dL, seizures, arrhythmia) | 3–4 amps Ca gluconate | Central line preferred; may require continuous infusion |
📌 Recheck serum calcium 4–6 hrs post-infusion
📌 Always correct magnesium if low
📌 Avoid mixing with phosphate or bicarbonate solutions
🛑 Critical Red Flags in Hypocalcemia
-
Check ionized calcium in alkalosis (may be falsely low total Ca)
-
Massive transfusion → citrate binds Ca²⁺ rapidly
-
Hypocalcemia + Hyperphosphatemia → avoid calcium until phosphate corrected
-
Monitor ECG closely: QT prolongation may precede torsades
-
Hypomagnesemia must be corrected first to avoid PTH resistance
🔥 Hypercalcemia (Ca²⁺ > 10.5 mg/dL or Ionized > 1.3 mmol/L)
🔎 Causes of Hypercalcemia
| Mechanism | Examples |
|---|---|
| Increased bone resorption | Hyperparathyroidism, malignancy (PTHrP, bone mets) |
| Increased gut absorption | Vitamin D toxicity, sarcoidosis |
| Decreased renal excretion | Thiazide diuretics, lithium |
| Prolonged immobilization | High bone turnover patients |
⚠️ Symptoms: "Stones, Bones, Groans, Thrones, Psychiatric Overtones"
-
Stones: Nephrolithiasis
-
Bones: Bone pain, fractures
-
Groans: Constipation, nausea, pancreatitis
-
Thrones: Polyuria, dehydration
-
Psychiatric: Confusion, lethargy, coma
💊 Management of Severe Hypercalcemia (>14 mg/dL or Symptomatic)
| Step | Therapy | Purpose |
|---|---|---|
| 1 | IV NS hydration | Expand ECF, promote calciuresis (200–300 mL/hr) |
| 2 | Loop diuretic (after hydration) | Enhance Ca excretion |
| 3 | Bisphosphonates (pamidronate, zoledronic acid) | Block bone resorption |
| 4 | Calcitonin | Rapid but short-acting effect |
| 5 | Dialysis | If refractory or renal failure |
🔎 Avoid thiazide diuretics — they increase calcium reabsorption.
🛑 Red Flags in Hypercalcemia
-
ECG: Short QT interval, risk of arrhythmia
-
Avoid aggressive correction unless symptomatic or Ca > 13–14 mg/dL
-
Malignancy-related hypercalcemia often needs bisphosphonates + hydration
-
Use calcitonin for acute drop while waiting for bisphosphonates to work
-
Hydration first, then diuretics
🧠 Clinical Pearls
-
Always check and correct magnesium in calcium disorders
-
Don't treat hypocalcemia based on total Ca alone — use ionized Ca or corrected formula
-
Albumin < 3 g/dL may significantly underestimate true calcium status
-
In the ICU, calcium chloride is reserved for cardiac arrest or severe, refractory cases — never give peripherally
4️⃣ Sodium Disorders – Hyponatremia & Hypernatremia in the ICU
🧪 Sodium (Na⁺) – Clinical Essentials
-
Normal Serum Na⁺: 135–145 mEq/L
-
Sodium is the primary extracellular cation and key determinant of plasma osmolality.
-
Regulated by:
-
ADH (vasopressin) – water retention
-
Thirst – water intake
-
Aldosterone – Na⁺ reabsorption in kidneys
-
📌 Sodium disorders are water problems, not just sodium problems.
🌡️ Serum Osmolality Equation
Osm = 2[Na⁺] + Glucose/18 + BUN/2.8
(Na⁺ in mEq/L, Glucose & BUN in mg/dL)
🧊 Hyponatremia (Na⁺ < 135 mEq/L)
✅ Stepwise Evaluation
-
Is it real?
-
Pseudohyponatremia from:
-
Severe hyperlipidemia
-
Hyperproteinemia (e.g., multiple myeloma)
-
Lab artifact
-
-
-
Check osmolality:
| Type | Osmolality | Example |
|---|---|---|
| Hypotonic | <275 mOsm/kg | True hyponatremia |
| Isotonic | 275–295 | Lab error, pseudohyponatremia |
| Hypertonic | >295 | Hyperglycemia, mannitol |
📌 Correct Na⁺ in hyperglycemia:
↓Na⁺ by 1.6 mEq/L for every 100 mg/dL ↑ glucose over 100
📊 Classify by Volume Status (Hypotonic Hyponatremia)
| Status | Causes | Urine Na⁺ |
|---|---|---|
| Hypovolemic | GI loss, diuretics, burns | <20 = extrarenal; >20 = renal loss |
| Euvolemic | SIADH, hypothyroid, adrenal insufficiency | >40 |
| Hypervolemic | CHF, cirrhosis, nephrotic syndrome | <20 |
🧠 SIADH = inappropriately concentrated urine in a euvolemic patient
⚠️ Symptoms of Hyponatremia
| Level (mEq/L) | Symptoms |
|---|---|
| 130–135 | Often asymptomatic |
| 125–129 | Nausea, headache, mild confusion |
| <120 | Seizures, coma, respiratory arrest |
🚨 Rapid drop → cerebral edema
⛑️ Treatment Principles
🔹 Rule of Safe Correction
Correct ≤ 8–10 mEq/L in 24h, ideally ≤ 6 mEq/L in high-risk patients
Too rapid = osmotic demyelination syndrome (ODS)
🔹 Treatment by Type
| Type | Fluid Choice |
|---|---|
| Hypovolemic | NS or balanced crystalloid |
| SIADH (euvolemic) | Fluid restriction, salt tabs, consider loop diuretics |
| Hypervolemic | Fluid + salt restriction, loop diuretics ± vasopressin antagonists |
💉 3% Hypertonic Saline – When & How
Indications:
-
Na⁺ <120 mEq/L with seizures, coma
-
ICP management (trauma, cerebral edema)
Dosing:
-
Bolus: 100 mL 3% NaCl over 10 minutes
-
Repeat up to 3 doses if needed
⚠️ ICU Box: Hypertonic Saline (3% NaCl) – Dosing & Correction Safety
🧂 Composition & Concentration
-
3% NaCl = 513 mEq/L of sodium
-
Therefore, 100 mL = 51.3 mEq Na⁺
🔢 A single 100 mL bolus can raise serum Na⁺ by 4–6 mEq/L
→ High risk for overshoot if not monitored
🛑 Correction Limits (24-Hour Max Sodium Rise)
| Patient Type | Max Safe Increase |
|---|---|
| High-Risk (malnourished, liver disease, alcoholism, elderly) | ≤6 mEq/L |
| Otherwise stable ICU patient | ≤8–10 mEq/L |
| Absolute max in 48 hrs | <18 mEq total |
💉 Emergency Protocol (Na⁺ <120 with Seizures or Coma)
-
Give 100 mL of 3% NaCl over 10 minutes
-
Recheck Na⁺ immediately
-
Repeat only as needed (max 3 times)
-
Target initial rise of +4–6 mEq/L, then slow correction
📌 Key ICU Notes:
-
Use central line if prolonged or continuous
-
Monitor Na⁺ every 2–4 hours during correction
-
Stop fluids or give DDAVP if sodium rises too fast
🔥 Hypernatremia (Na⁺ > 145 mEq/L)
🔎 Causes by Volume Status
| Status | Common Causes |
|---|---|
| Hypovolemic | GI loss, osmotic diuresis, burns |
| Euvolemic | Diabetes insipidus (central or nephrogenic) |
| Hypervolemic | Hypertonic saline, bicarbonate infusion, Cushing’s |
⚠️ Symptoms
-
Lethargy, irritability
-
Seizures, coma
-
Dehydration signs (dry mucosa, hypotension)
-
Brain shrinkage → vascular rupture, ICH
🧮 Free Water Deficit Formula
Deficit (L) = TBW × [(Na⁺ / 140) − 1]
TBW = 0.6 × weight (kg) in men, 0.5 in women
💧 Treatment Principles
-
Use hypotonic fluids:
-
D5W (no sodium)
-
½ NS (0.45% NaCl)
-
-
Calculate deficit and replace over 48–72 hours
-
Max correction = ≤10–12 mEq/day
📌 Too fast correction → cerebral edema
💉 Diabetes Insipidus (Central vs. Nephrogenic)
| Type | ADH | Urine Osm | Rx |
|---|---|---|---|
| Central DI | ↓ | <300 | Desmopressin |
| Nephrogenic DI | Normal | <300 | Thiazides, NSAIDs, low Na⁺ diet |
🧠 Clinical Pearls
-
Never give free water IV directly — use D5W or enteral
-
3% NaCl is a rescue drug — treat with discipline
-
Always calculate and monitor free water deficit in hypernatremia
-
In DI, urine output may exceed 4–5 L/day — replace cautiously
-
Sodium derangements are fluid logic puzzles — not isolated lab values
5️⃣ Magnesium Disorders – ICU Management of Hypo- & Hypermagnesemia
🧬 Magnesium (Mg²⁺) – The Forgotten Stabilizer
-
Normal serum range: 1.5–2.5 mEq/L (0.75–1.25 mmol/L)
-
99% is intracellular or bone-bound
-
Only ~1% is in plasma — poor reflection of total stores
📌 Key Physiological Roles
-
ATP synthesis (Mg-ATP is the active form)
-
Cardiac conduction and myocardial stabilization
-
Neuromuscular excitability and reflex control
-
Cofactor for Na⁺/K⁺ ATPase, PTH secretion, insulin function
-
Regulates K⁺ and Ca²⁺ channels
📍 Without magnesium, potassium and calcium won't correct — this is an ICU red flag.
🔻 Hypomagnesemia (Mg²⁺ < 1.5 mEq/L)
🔎 Causes of Hypomagnesemia
| Mechanism | Examples |
|---|---|
| GI Loss | Diarrhea, vomiting, fistulas, malabsorption, NG suction |
| Renal Loss | Diuretics (loop > thiazide), aminoglycosides, cisplatin, amphotericin |
| Alcoholism | Poor intake + increased renal loss |
| Refeeding Syndrome | Shifts Mg²⁺ into cells |
| DKA Treatment | Insulin drives Mg²⁺ into cells |
| Pancreatitis, Sepsis | Increased demand or redistribution |
⚠️ Clinical Features
-
Neuromuscular:
-
Tremors, weakness, cramps
-
Hyperreflexia
-
Seizures (esp. in children)
-
-
Cardiac:
-
Torsades de pointes
-
Prolonged QT, PR, and QRS
-
-
Often coexists with hypokalemia or hypocalcemia
💉 IV Magnesium Replacement in the ICU
| Formulation | MgSO₄ Content | Mg²⁺ (mEq) |
|---|---|---|
| 1 g MgSO₄ | = 8 mEq Mg²⁺ | in 2 mL ampoule (50%) |
| 2 g MgSO₄ | = 16 mEq Mg²⁺ | in 4–10 mL volume |
📋 Replacement Protocol (Stepwise)
| Severity | Dose | Route |
|---|---|---|
| Mild (1.2–1.5 mEq/L) | 2 g MgSO₄ IV over 2 hrs | Peripheral |
| Moderate (1.0–1.2) | 4 g MgSO₄ IV over 4 hrs | Prefer central |
| Severe (<1.0 or torsades) | 2 g IV push over 10 min, repeat x2 | Central line or monitored setting |
🔁 Follow with infusion of 4–6 g MgSO₄ over 12–24 hours
🛑 Safety Tips
-
Avoid rapid push unless cardiac arrest or torsades
-
Monitor deep tendon reflexes and respiratory status
-
Recheck serum Mg²⁺ every 6–12 hours
-
Check renal function — reduce dose if GFR <30 mL/min
📌 Clinical Clue
If hypokalemia is not correcting, and you've already replaced K⁺ — always replace Mg²⁺ first.
🔺 Hypermagnesemia (Mg²⁺ > 2.5 mEq/L)
🔎 Causes
| Source | Example |
|---|---|
| Excess intake | Mg antacids, enemas in renal failure |
| Renal failure | ↓ Mg excretion |
| Iatrogenic | High-dose MgSO₄ for preeclampsia |
| Adrenal insufficiency | Rare cause via volume depletion |
⚠️ Symptoms by Level
| Mg²⁺ Level (mEq/L) | Clinical Signs |
|---|---|
| 3.5–5 | Nausea, flushing, hypotension |
| 5–7 | Loss of DTRs, bradycardia |
| 7–10 | Somnolence, heart block |
| >10 | Paralysis, respiratory arrest, cardiac arrest (asystole) |
🛑 Emergency Management of Severe Hypermagnesemia
| Step | Action |
|---|---|
| 1️⃣ | Stop all Mg sources immediately |
| 2️⃣ | Give 10 mL of 10% calcium gluconate IV over 10 min |
| 3️⃣ | Start IV fluids + loop diuretics (if renal function intact) |
| 4️⃣ | Consider dialysis if: |
-
Oliguria/anuria
-
Mg²⁺ > 8 mEq/L with symptoms
💊 Calcium as the Antagonist
-
Calcium antagonizes Mg at neuromuscular junctions
-
Always use gluconate unless central line access is available
💡 Clinical Pearls
-
Mg²⁺ is a hidden electrolyte — low with little lab warning
-
In torsades de pointes, 2 g IV MgSO₄ is the drug of choice
-
In hypokalemia or hypocalcemia, always check magnesium
-
Magnesium replacement is safe via peripheral line up to 2 g/hr
-
In preeclampsia → monitor DTRs, RR, and UOP hourly
6️⃣ Phosphate & Chloride Disorders – ICU Implications & Management
🔬 Phosphate (PO₄³⁻) – Energy, Muscle, and Bone
-
Normal serum phosphate: 2.5–4.5 mg/dL (0.81–1.45 mmol/L)
-
85% in bone, 14% intracellular, <1% in plasma
-
Essential for:
-
ATP, 2,3-DPG (oxygen release from hemoglobin)
-
Muscle contractility (especially diaphragm)
-
Leukocyte and platelet function
-
Buffering H⁺ in acid-base homeostasis
-
🔻 Hypophosphatemia (PO₄³⁻ < 2.5 mg/dL)
🔎 Common ICU Causes
| Mechanism | Example |
|---|---|
| Redistribution | Refeeding syndrome, insulin, respiratory alkalosis |
| Increased excretion | Diuretics, DKA recovery, hyperparathyroidism |
| Decreased intake | Malnutrition, alcoholism, TPN without phosphate |
| Binding | Antacids, sepsis, catecholamine surges |
⚠️ Clinical Consequences
-
Weakness, myopathy → weaning failure from ventilator
-
Encephalopathy, seizures
-
↓ Myocardial contractility → hypotension
-
Hemolysis (ATP depletion in RBCs)
-
↓ 2,3-DPG → tissue hypoxia
📌 Symptoms usually appear when PO₄³⁻ < 1.0 mg/dL
💉 Replacement Strategy – Stepwise
| Severity | IV Dose | Route |
|---|---|---|
| Mild (2.0–2.5) | Oral phosphate salts | PO if tolerated |
| Moderate (1.0–2.0) | 0.16–0.32 mmol/kg | Slow IV |
| Severe (<1.0 or symptomatic) | 0.64 mmol/kg | IV over 6–8 hrs |
💉 IV Phosphate Preparations
| Agent | Phosphate Content |
|---|---|
| NaPO₄ | 3 mmol/mL |
| KPO₄ | 3 mmol/mL |
💡 Choose Na⁺ or K⁺ salt depending on concurrent electrolyte levels
📌 Always dilute and infuse over 4–6 hours
📌 Recheck PO₄³⁻ after 6–12 hours
🛑 Red Flags in Repletion
-
Rapid IV infusion → hypocalcemia or calcium-phosphate precipitation
-
In renal failure: reduce dose by 50–75%
-
If PO₄³⁻ < 1.0 mg/dL, ensure Mg²⁺ is corrected first
🔺 Hyperphosphatemia (PO₄³⁻ > 4.5 mg/dL)
🔎 Causes
| Mechanism | Example |
|---|---|
| Renal failure | ↓ PO₄³⁻ excretion |
| Cell lysis | Tumor lysis, rhabdomyolysis |
| Acidosis | Shifts phosphate out of cells |
| Excess intake | Phosphate enemas, supplements |
⚠️ Risks
-
Calcium phosphate precipitation → hypocalcemia, soft tissue calcification
-
AKI and worsening of secondary hyperparathyroidism
-
ECG: prolonged QT if hypocalcemia is induced
🛠️ Management
-
IV fluids to promote renal excretion
-
Phosphate binders:
-
Calcium acetate
-
Sevelamer
-
-
Dialysis if severe or in ESRD
🧪 Chloride (Cl⁻) – The Acid-Base Shadow Player
🔎 Normal Range: 96–106 mEq/L
Chloride is the primary extracellular anion, often passive but key in:
-
Acid-base balance (via strong ion difference)
-
Maintenance of electroneutrality
-
Renal HCO₃⁻ exchange
-
Influence on AG and SID (Stewart approach)
🔻 Hypochloremia (Cl⁻ < 96 mEq/L)
🧠 Causes
-
Vomiting, NG suction (loss of HCl)
-
Loop diuretics
-
Dilutional (fluid overload, CHF)
-
Metabolic alkalosis
⚠️ Clues
-
Metabolic alkalosis with high bicarbonate
-
High urine chloride = renal cause
-
Low urine chloride = extrarenal loss
💉 Management
-
Replace with NS (154 mEq/L Cl⁻)
-
Stop volume depletion triggers (diuretics)
-
In alkalosis: consider acetazolamide
🔺 Hyperchloremia (Cl⁻ > 106 mEq/L)
🧠 Causes
-
0.9% NaCl overuse → non-anion gap acidosis
-
Renal tubular acidosis (Type 1 & 4)
-
High chloride-containing meds or nutrition
⚠️ Effects
-
Vasoconstriction → ↓ renal perfusion
-
↓ bicarbonate (hyperchloremic metabolic acidosis)
-
Confusion, fatigue if severe
🛠️ Management
-
Switch fluids: NS → balanced crystalloids (e.g., PlasmaLyte, LR)
-
Treat acidosis if symptomatic
-
Adjust TPN or medications
🧠 Clinical Pearls
-
Hypophosphatemia = ventilator weaning failure until corrected
-
Phosphate binds free calcium → be cautious in refeeding & renal failure
-
Hyperchloremia is iatrogenic — avoid NS overload
-
For metabolic alkalosis with hypochloremia, NS is the therapy
-
Always interpret chloride in relation to acid-base and sodium status
7️⃣ Combined Electrolyte Disturbances – ICU Patterns, Syndromes & Strategy
🧠 Why Think in Clusters?
In critical care, electrolyte disorders rarely occur alone. They often represent underlying pathology, organ dysfunction, or iatrogenic triggers.
Understanding electrolyte triads and paired abnormalities allows you to:
🔹 Predict lab shifts before they occur
🔹 Prioritize treatment order (e.g., Mg²⁺ before K⁺)
🔹 Avoid futile replacement
🔹 Spot systemic syndromes early (e.g., refeeding, tumor lysis)
🔺 The Classic Triads & Clinical Clues
🧩 1. K⁺ – Mg²⁺ – Ca²⁺ Triad
| Pattern | Likely Scenario | Clinical Tip |
|---|---|---|
| ↓ K⁺ + ↓ Mg²⁺ + ↓ Ca²⁺ | Alcoholism, diarrhea, diuretics, sepsis | Replace Mg²⁺ first to fix others |
| ↓ Ca²⁺ + ↓ Mg²⁺ | PPI use, sepsis, pancreatitis | Check ionized Ca and Mg; may need IV gluconate |
| ↓ K⁺ despite K⁺ replacement | DKA, diarrhea, cisplatin | Hidden culprit = low Mg²⁺ |
🧩 2. PO₄³⁻ – K⁺ – Mg²⁺ in Refeeding Syndrome
Occurs in malnourished, alcoholic, or post-ICU fasting patients when calories or glucose are reintroduced.
| Early Sign | ↓ PO₄³⁻ |
|---|---|
| Followed by | ↓ Mg²⁺, ↓ K⁺, edema, CHF, arrhythmias |
📌 Prevent by prophylactic repletion and slow feeding (<20 kcal/kg/day initially)
🧩 3. DKA Pattern: K⁺ and PO₄³⁻
-
Initial labs may show high or normal K⁺, normal phosphate
-
But after insulin:
-
Rapid intracellular shift → ↓ K⁺, ↓ PO₄³⁻, ↓ Mg²⁺
-
📌 Monitor electrolytes q4–6h
📌 Replace K⁺ early if <5.3 even if "normal"
🧩 4. Tumor Lysis Syndrome (TLS)
| Electrolyte | Trend |
|---|---|
| K⁺ | ↑ |
| PO₄³⁻ | ↑ |
| Ca²⁺ | ↓ (due to phosphate binding) |
| Uric acid | ↑ |
| Mg²⁺ | ↓ (from Ca precipitation or binding) |
📌 Leads to renal failure, arrhythmia, seizures
📌 Use aggressive hydration, allopurinol, and phosphate binders
🧩 5. Diarrhea Syndrome
| Lost in stool | HCO₃⁻, K⁺, Mg²⁺, PO₄³⁻ |
| Consequences | Metabolic acidosis, dehydration, muscle weakness |
| Labs | ↓ K⁺, ↓ Mg²⁺, ↓ PO₄³⁻, non-anion gap acidosis |
📌 Use balanced fluids, avoid NS overload
📌 Replace electrolytes sequentially (Mg²⁺ first)
🧩 6. Massive Transfusion Syndrome
| Lost or diluted | Ionized Ca²⁺, Platelets, Fibrinogen |
| Mechanism | Citrate binds calcium, massive dilution |
| Effect | Hypotension, bleeding, prolonged QT |
📌 Replace calcium gluconate IV every 4–6 units PRBC
📌 Monitor ionized Ca²⁺, not total Ca
🚨 Electrolyte Red Flags in ICU
| Scenario | What to Do |
|---|---|
| Hypokalemia unresponsive to K⁺ | Check and replace Mg²⁺ |
| Hypocalcemia + hyperphosphatemia | Delay Ca²⁺ → correct PO₄³⁻ first |
| Aggressive insulin therapy | Monitor K⁺ and PO₄³⁻ every 4–6 hrs |
| NG suction / vomiting | Replace Cl⁻ + K⁺, not just volume |
| Rapid Na⁺ correction | Beware ODS → slow and monitor q2–4 hrs |
| Volume overload + hyperchloremia | Switch to balanced fluids (LR or Plasma-Lyte) |
📘 High-Yield ICU Syndromes Summary
| Syndrome | Signature Labs | Core Treatment |
|---|---|---|
| Refeeding | ↓ K⁺, ↓ Mg²⁺, ↓ PO₄³⁻ | Replete first, feed slow |
| TLS | ↑ K⁺, ↑ PO₄³⁻, ↓ Ca²⁺ | Hydration + rasburicase/allopurinol |
| DKA | Initial ↑ K⁺, later ↓ K⁺/Mg²⁺/PO₄³⁻ | Monitor & replete early |
| Massive transfusion | ↓ ionized Ca²⁺ | IV Ca gluconate |
| TPN-associated | ↓ Mg²⁺, ↓ PO₄³⁻ | Add to TPN or replace IV |
| Chronic diarrhea | ↓ K⁺, ↓ Mg²⁺, ↓ HCO₃⁻ | Oral or IV replacement |
8️⃣ ICU Replacement Strategies – Daily Needs, Infusion Limits & Compatibility Tables
🩺 Daily Maintenance Requirements (70 kg Adult)
| Electrolyte | Daily Need | Comment |
|---|---|---|
| Sodium (Na⁺) | 1–2 mEq/kg/day → 70–140 mEq | Adjust in hyponatremia, losses |
| Potassium (K⁺) | 1 mEq/kg/day → 70 mEq | Add deficit if <3.5 mEq/L |
| Calcium (Ca²⁺) | 1,000–1,200 mg (elemental) | Use corrected calcium |
| Magnesium (Mg²⁺) | 8–20 mEq/day | Higher in ICU/sepsis |
| Phosphate (PO₄³⁻) | 20–40 mmol/day | Replace in DKA, refeeding |
| Chloride (Cl⁻) | Usually coupled with Na⁺/K⁺ | Watch for hyperchloremic acidosis |
🧮 Deficit Correction Quick Formulas
| Electrolyte | Formula | Units |
|---|---|---|
| K⁺ | 0.3 × kg × (3.5 − K⁺) | mEq |
| Ca²⁺ (corrected) | Measured Ca + 0.8 × (4 − albumin) | mg/dL |
| PO₄³⁻ | 0.32 mmol/kg for moderate; 0.64 mmol/kg for severe | mmol |
| Na⁺ (free water) | TBW × [(Na⁺/140) − 1] | Liters of water deficit |
| Mg²⁺ | Empiric: 2–4 g (16–32 mEq/day) | IV g/day |
📌 TBW = 0.6 × weight (kg) in males, 0.5 in females
💧 Maximum Infusion Rates – Central vs. Peripheral
| Electrolyte | Max Rate Peripheral | Max Rate Central | Comments |
|---|---|---|---|
| K⁺ | 10–20 mEq/hr | 40 mEq/hr | Monitor ECG; central line >20 mEq/hr |
| Mg²⁺ | 1 g/hr (8 mEq) | 2 g/hr | Faster in torsades or eclampsia |
| Ca²⁺ (gluconate) | 1 amp over 10–20 min | Multiple amps in central | Avoid mixing with bicarbonate/phosphate |
| PO₄³⁻ | 7.5–15 mmol over 4–6 hrs | Up to 30 mmol | Use Na or K salt accordingly |
| 3% NaCl | Not recommended | 100 mL over 10 min | Seizures/Na⁺ <120; monitor every 2h |
🔀 When to Choose Na⁺ vs. K⁺ Phosphate or Bicarbonate Salt
| Condition | Use |
|---|---|
| Hypophosphatemia + Hypokalemia | Use KPO₄ |
| Hypophosphatemia + Hyperkalemia | Use NaPO₄ |
| Metabolic acidosis + Hypokalemia | Use KHCO₃ (carefully!) |
| Volume overload + Hyperchloremia | Avoid NaCl; use balanced solutions |
🧪 IV Compatibility Quick Table
| Do NOT Mix Together | Reason |
|---|---|
| Calcium + Phosphate | Precipitates in blood & line |
| Calcium + Bicarbonate | White precipitate (calcium carbonate) |
| K⁺ + Dextrose alone | May mask hypokalemia |
| Phosphate + Mg²⁺ (in same line) | Risk of precipitate, clog |
| Magnesium + Rapid IV push | Bradycardia, respiratory depression |
📦 Electrolyte Ampoule Summary (Standard ICU Supply)
| Drug | Volume | Approx. Electrolyte |
|---|---|---|
| KCl 7.5% | 10 mL | 10 mEq |
| KCl 10% | 10 mL | 13.4 mEq |
| KCl 15% | 10 mL | 20 mEq |
| MgSO₄ 50% | 2 mL | 1 g = 8 mEq |
| Ca gluconate 10% | 10 mL | ~90–100 mg elemental Ca²⁺ |
| NaPO₄ / KPO₄ | 1 mL | 3 mmol phosphate |
| 3% NaCl | 100 mL | 51.3 mEq Na⁺ |
📍 Clinical Pearls in Repletion Strategy
-
K⁺, Mg²⁺, PO₄³⁻ = replete in that order
-
Replace Mg²⁺ first in any refractory hypokalemia or hypocalcemia
-
Always use central access for high-concentration or high-rate infusions
-
Avoid using NS alone in hyperchloremic acidosis — switch to PlasmaLyte or LR
-
In AKI/CKD patients → reduce Mg²⁺, PO₄³⁻, K⁺ doses or dialyze.
9️⃣ Electrolytes & Acid-Base Interaction – Understanding the Hidden Ties
🧪 The Core Principle
Electrolyte imbalances do not happen in isolation — they influence or reflect acid-base disturbances. Some shifts are compensatory, others are pathological. Recognizing this interplay is crucial for correct diagnosis and targeted therapy.
🔄 Potassium (K⁺) and Acid-Base Status
| Acid-Base State | K⁺ Shift |
|---|---|
| Metabolic Acidosis | K⁺ shifts OUT of cells → hyperkalemia |
| Alkalosis | K⁺ shifts INTO cells → hypokalemia |
🔹 But this shift is transcellular, not total-body.
🔹 Patients may have normal serum K⁺ but be profoundly depleted intracellularly.
📌 In DKA, insulin + acidosis correction → K⁺ plummets
📌 Replace K⁺ aggressively once insulin started, even if initial K⁺ is normal
🌬️ Chloride (Cl⁻) and Bicarbonate (HCO₃⁻)
Chloride is a strong anion that directly influences HCO₃⁻ via exchange in the renal tubule:
-
Hyperchloremia → ↓ HCO₃⁻ → non-anion gap metabolic acidosis
-
Hypochloremia → ↑ HCO₃⁻ → metabolic alkalosis
🔬 Chloride & Fluid Choice
| Fluid | Cl⁻ Content | Acid-Base Effect |
|---|---|---|
| 0.9% NaCl | 154 mEq/L | May cause acidosis |
| PlasmaLyte / LR | ~98–110 mEq/L | More balanced, less acidosis risk |
📌 In resuscitation, excessive NS → hyperchloremic metabolic acidosis
⚠️ Albumin & Anion Gap (AG)
-
Albumin is a major unmeasured anion
-
↓ Albumin → ↓ Anion Gap
(Masked high AG acidosis)
Corrected AG = AG + (2.5 × [4 − albumin])
✅ Use this in sepsis, burns, liver failure
✅ Otherwise, you may miss lactic acidosis or DKA
📈 Phosphate & pH
-
PO₄³⁻ acts as a buffer, especially in renal tubules
-
In alkalosis, intracellular PO₄³⁻ uptake ↑ → serum phosphate ↓
-
In DKA or lactic acidosis, phosphate is lost in urine and shifts intracellularly with insulin
📌 Monitor PO₄³⁻ closely in alkalosis, sepsis, and refeeding
⚙️ Magnesium & pH
-
Alkalosis may decrease ionized Mg²⁺, mimicking hypomagnesemia
-
Hypomagnesemia exacerbates alkalosis by impairing renal K⁺ and Ca²⁺ handling
-
Watch in:
-
Sepsis
-
Diuretic overuse
-
Massive transfusion
-
🧠 Stewart’s Strong Ion Concept (Simplified)
| Variable | Impact |
|---|---|
| ↑ Na⁺ – Cl⁻ gap (strong ion difference) | ↑ HCO₃⁻ → alkalosis |
| ↓ Na⁺ – Cl⁻ gap | ↓ HCO₃⁻ → acidosis |
So:
-
Giving NS (Cl⁻ rich) narrows SID → acidosis
-
Balanced fluids preserve SID → stable pH
🔺 Clinical Summary Table
| Electrolyte Shift | Acid-Base Effect |
|---|---|
| ↑ K⁺ (acidosis) | Due to H⁺ shift into cells |
| ↓ K⁺ (alkalosis) | H⁺ out, K⁺ into cells |
| ↓ Cl⁻ | ↑ HCO₃⁻ = alkalosis |
| ↑ Cl⁻ | ↓ HCO₃⁻ = acidosis |
| ↓ Albumin | Lowers AG → may mask high AG acidosis |
| ↓ PO₄³⁻ | Seen in alkalosis and insulin therapy |
| ↓ Mg²⁺ | Aggravates alkalosis and refractory K⁺/Ca²⁺ loss |
💡 Clinical Pearls
-
Never interpret K⁺ without pH
-
Chloride drives acid-base in saline vs. balanced fluid resuscitation
-
Use corrected AG in hypoalbuminemia
-
PO₄³⁻ and Mg²⁺ derangements may be the silent culprits in acid-base failures
-
In persistent metabolic alkalosis → check for Cl⁻ loss and volume depletion
🔟 Clinical Scenarios + Red Flag Tips – Real ICU Cases from the Electrolyte Frontlines
Each of these scenarios is based on real ICU patterns, designed to reinforce decision-making, replacement strategies, and the clinical judgment needed in moments of silence and instability.
🧠 Scenario 1: Refractory Hypokalemia in a Diabetic Patient
🧾 A 55-year-old man with DKA is on insulin. Serum K⁺ is 3.3 despite receiving 80 mEq over 12 hours. Mg²⁺ = 1.1 mEq/L.
✅ Diagnosis: Uncorrected hypomagnesemia
🔺 Red Flag: Hypokalemia that doesn't correct → replace Mg²⁺ first
📌 Give 2–4 g MgSO₄ IV, then resume K⁺ replacement
⚠️ Scenario 2: Confusion After Rapid Sodium Correction
🧾 72-year-old woman with chronic hyponatremia (Na⁺ 112). Given 3 boluses of 3% NaCl in 6 hours. Repeat Na⁺ = 124. She becomes confused, then obtunded.
✅ Diagnosis: Overcorrection → Osmotic Demyelination Syndrome (ODS)
🔺 Red Flag: Na⁺ ↑ more than 10 mEq/24 hrs
📌 Stop hypertonic saline
📌 Give D5W + Desmopressin (DDAVP) to slow the rise
🧠 Scenario 3: Weakness & QT Prolongation After Transfusion
🧾 Massive transfusion protocol (6 units PRBC). BP drops, ECG shows QTc 510 ms. Ionized Ca²⁺ = 0.9 mmol/L.
✅ Diagnosis: Citrate-induced hypocalcemia
🔺 Red Flag: Every 4 units of blood = 1 amp Ca gluconate
📌 Give 10 mL of 10% Ca gluconate IV slowly
📌 Recheck ionized calcium, monitor ECG
⚠️ Scenario 4: Seizures on POD#1 with Normal Labs
🧾 Post-op man with ileus and vomiting. Labs: Na⁺ = 137, Ca²⁺ = 8.7, K⁺ = 3.8. PO₄³⁻ = 0.6 mg/dL. Mg²⁺ = 1.0.
✅ Diagnosis: Severe hypophosphatemia + hypomagnesemia
🔺 Red Flag: Serum phosphate <1.0 mg/dL → seizures, diaphragmatic weakness
📌 Replace with 15–30 mmol IV PO₄³⁻ over 6 hours
📌 Replete Mg²⁺ simultaneously
🧠 Scenario 5: Hyperchloremic Acidosis After Resuscitation
🧾 Young trauma patient received 5 L NS. Now pH = 7.28, HCO₃⁻ = 17, Cl⁻ = 114, AG = normal
✅ Diagnosis: Non-anion gap metabolic acidosis due to hyperchloremia
🔺 Red Flag: NS >3–4 L → ↑ Cl⁻ → ↓ HCO₃⁻
📌 Switch to PlasmaLyte or LR
📌 Give bicarbonate only if pH <7.2 or symptomatic
⚠️ Scenario 6: Refeeding Syndrome on Day 3 of ICU Stay
🧾 ICU patient with chronic alcoholism started on TPN. Labs show ↓ K⁺ = 2.8, ↓ Mg²⁺ = 1.0, ↓ PO₄³⁻ = 0.7
✅ Diagnosis: Refeeding syndrome
🔺 Red Flag: Start feeds slowly (<20 kcal/kg/day) + prophylactic Mg²⁺, K⁺, PO₄³⁻
📌 Hold feeding temporarily
📌 Replace all 3 electrolytes IV
🧠 Scenario 7: Respiratory Arrest in a Pregnant Woman on Magnesium Drip
🧾 Eclampsia patient received 6 g MgSO₄ bolus + 2 g/hr infusion. Now RR = 6, DTRs absent, Mg²⁺ = 8.5 mEq/L
✅ Diagnosis: Iatrogenic hypermagnesemia
🔺 Red Flag: Toxic Mg²⁺ level > 6 mEq/L → ↓ DTRs, then ↓ RR, then cardiac arrest
📌 Stop MgSO₄
📌 Give IV Ca gluconate 10 mL of 10% to antagonize
⚠️ Scenario 8: Bradycardia & Paralysis in a Renal Patient
🧾 ESRD patient accidentally given Mg-containing laxatives. Mg²⁺ = 7.2 mEq/L. ECG: bradycardia, 2nd-degree AV block
✅ Diagnosis: Severe hypermagnesemia with neuromuscular toxicity
🔺 Red Flag: Renal failure + Mg supplements = toxic
📌 Give IV Ca gluconate
📌 Start dialysis if symptomatic or Mg²⁺ > 8
📍 10 ICU Red Flags – Quick Review Table
| Red Flag | Action |
|---|---|
| Na⁺ correction >10 mEq/day | Risk of ODS → stop fluids, give DDAVP |
| K⁺ not correcting | Check Mg²⁺ — replace it first |
| Mg²⁺ >6 with RR ↓ or DTRs absent | Stop Mg, give Ca gluconate |
| Citrate transfusion (↓ Ca²⁺) | Give Ca gluconate after 4 units PRBC |
| PO₄³⁻ <1.0 mg/dL | IV phosphate urgently |
| NS >4 L → Cl⁻ 114 | Switch to PlasmaLyte, risk of acidosis |
| Hypochloremia + alkalosis | NS resuscitation corrects it |
| Refeeding labs drop | Hold feeds, replete Mg/K/PO₄³⁻ first |
| Na⁺ rises too fast in seizures | Stop fluids, consider D5W + DDAVP |
| Ca + PO₄³⁻ infused together | Risk of precipitation → avoid mixing |
1️⃣1️⃣ Resource-Limited Practice Essentials – ICU Electrolyte Care Without the Luxury
In many parts of the world, central lines, pumps, ionized calcium, or 24/7 labs may be unavailable. But smart bedside practice can still achieve safe, effective electrolyte correction — with judgment, timing, and adaptation.
🧪 General Principles for Low-Resource Electrolyte Replacement
✅ Treat clinically, not just by numbers
✅ Prefer oral or enteral routes when possible
✅ Use diluted IV solutions and gravity drip with timed boluses
✅ Repeat doses only when there’s:
-
Clinical improvement
-
Urine output
-
No ECG red flags
🧴 Dilution Tables for Safe Peripheral Use (No Infusion Pump)
| Drug | Safe Dilution | Drip Rate (approx.) |
|---|---|---|
| KCl 10 mEq | in 500 mL NS | over 2–4 hrs |
| MgSO₄ 2 g (16 mEq) | in 100 mL D5W | over 30–60 min |
| Ca gluconate 1 amp | in 50–100 mL NS | over 15–30 min |
| NaPO₄ 15 mmol | in 250 mL NS | over 4 hrs |
| 3% NaCl (100 mL) | undiluted over 10 min | for seizures only |
📌 Use gravity drip chamber or micro-dripper sets
📌 Count drops per minute if no electronic regulation
🧠 Red Flag Protocols Without Monitors
| Condition | Bedside Rule |
|---|---|
| K⁺ Replacement | Ensure urine output >40 mL/hr before giving |
| Ca gluconate | Give 1 amp slowly over 20 min if arrhythmia, tetany, or transfusion |
| MgSO₄ | 2 g IV over 30 min → observe reflexes, RR hourly |
| PO₄³⁻ | If severe weakness, weaning failure, or hemolysis → 15 mmol IV PO₄³⁻ in 250 mL slowly |
🔄 Oral Repletion When IV Is Not Available
| Electrolyte | Oral Agent | Comment |
|---|---|---|
| K⁺ | KCl syrup 10% (20 mEq/15 mL) | Divide into 3 doses/day; mix with juice |
| Mg²⁺ | Magnesium oxide or syrup | Titrate to prevent diarrhea |
| Ca²⁺ | Calcium carbonate 500–1000 mg BID | Prefer chewable or powdered |
| PO₄³⁻ | Oral sodium phosphate solution | Used in bowel prep; dilute well |
📌 Divide doses, especially in elderly or GI sensitive patients
📌 Dilute in juice or ORS to reduce irritation
🩺 No Labs? Use Clinical Markers
| Sign | Likely Deficiency |
|---|---|
| Muscle cramps + arrhythmia | K⁺, Mg²⁺ |
| Tetany + prolonged QT | Ca²⁺ |
| Failure to wean from ventilator | PO₄³⁻ |
| Bradycardia + DTR loss | Mg²⁺ excess |
| Recurrent seizures in malnutrition | PO₄³⁻, Mg²⁺ |
💡 Local Hacks That Save Lives
✅ Mix KCl ampoule into 1L NS and mark the time — divide into 4 hourly doses
✅ Use drop count method:
-
~20 drops = 1 mL (macro set)
-
~60 drops = 1 mL (micro set)
✅ Keep ORS sachets + sugar-salt solutions available
✅ Use antacid suspensions for calcium orally
✅ Train staff to watch reflexes and breathing, not just numbers
❤️ Clinical Pearl for Resource-Limited Settings
“Where there is no monitor, your eyes become the monitor.
Where there is no lab, your clinical signs become the test.
Where there is no pump, your time becomes the regulation.”
– For every rural ICU, nurse, and technician rising with science
1️⃣2️⃣MCQs – Electrolyte Mastery Questions (With Answers & Explanations)
1. A 70 kg ICU patient has serum potassium of 2.6 mEq/L. What is the approximate total K⁺ deficit?
A. 30 mEq
B. 60 mEq
C. 100 mEq
D. 210 mEq ✅
Explanation:
Deficit = 0.3 × 70 × (3.5 − 2.6) = 18.9 × 3 = ~56.7 + maintenance (70) ≈ 130–150 → round to ~210 mEq total
2. Which electrolyte abnormality must be corrected before potassium or calcium will normalize?
A. Chloride
B. Bicarbonate
C. Magnesium ✅
D. Phosphate
Explanation:
Magnesium is a cofactor for Na⁺/K⁺ ATPase and PTH release. Its deficiency causes refractory hypokalemia and hypocalcemia.
3. What is the maximum safe peripheral infusion rate for K⁺?
A. 10 mEq/hr ✅
B. 20 mEq/hr
C. 40 mEq/hr
D. 60 mEq/hr
Explanation:
Peripheral IV: ≤10 mEq/hr (20 only with excellent access); faster rates require central line and ECG monitoring.
4. Which lab pattern suggests refeeding syndrome?
A. ↑ Na⁺, ↑ Mg²⁺, ↑ PO₄³⁻
B. ↓ K⁺, ↓ Mg²⁺, ↓ PO₄³⁻ ✅
C. ↓ Ca²⁺, ↑ PO₄³⁻, ↑ Mg²⁺
D. ↑ Cl⁻, ↑ HCO₃⁻, ↓ K⁺
Explanation:
Classic refeeding = rapid intracellular shifts → ↓ K⁺, ↓ Mg²⁺, ↓ phosphate
5. In tumor lysis syndrome, what electrolyte pattern is most expected?
A. ↓ K⁺, ↓ PO₄³⁻, ↓ uric acid
B. ↑ K⁺, ↑ PO₄³⁻, ↓ Ca²⁺ ✅
C. ↑ Ca²⁺, ↓ PO₄³⁻, ↓ Mg²⁺
D. ↓ K⁺, ↑ Ca²⁺, ↑ phosphate
Explanation:
Massive cell lysis releases K⁺ and phosphate → phosphate binds Ca²⁺ → hypocalcemia
6. Which of the following is a correct IV content match?
A. 10% Ca gluconate = 270 mg elemental calcium
B. 10% Ca chloride = 90 mg elemental calcium
C. 1 g MgSO₄ = 8 mEq magnesium ✅
D. 10 mL 7.5% KCl = 13.4 mEq potassium
Explanation:
1 g MgSO₄ = 8 mEq; Ca chloride has 270 mg, gluconate only 90–100 mg.
7. What is the free water deficit in a 60 kg man with Na⁺ of 158?
A. 1.5 L
B. 2.7 L ✅
C. 3.8 L
D. 4.5 L
Explanation:
TBW = 0.6 × 60 = 36
Deficit = 36 × [(158/140) − 1] ≈ 2.7 L
8. Giving 3% NaCl 100 mL over 10 minutes raises serum Na⁺ by about:
A. 1–2 mEq/L
B. 3–4 mEq/L
C. 4–6 mEq/L ✅
D. 8–10 mEq/L
Explanation:
100 mL = 51.3 mEq; may increase Na⁺ by 4–6 mEq/L, enough to treat seizures, but risks ODS if uncontrolled.
9. Which chloride level is most likely associated with normal anion gap metabolic acidosis?
A. 96
B. 102
C. 106
D. 114 ✅
Explanation:
Hyperchloremia (Cl⁻ > 110) often causes non-anion gap metabolic acidosis, especially after large NS resuscitation.
10. Which is most accurate regarding phosphate replacement?
A. Oral preferred when phosphate <1 mg/dL
B. Use KPO₄ if patient has hyperkalemia
C. Severe hypophosphatemia may impair diaphragm function ✅
D. Phosphate and calcium can be mixed safely in same line
Explanation:
PO₄³⁻ <1 → ATP depletion → muscle weakness, ventilator weaning failure
11. A patient receiving massive transfusion becomes hypotensive. ECG shows QT prolongation. Which is best initial treatment?
A. IV magnesium
B. Calcium gluconate ✅
C. Sodium bicarbonate
D. Fluid bolus
Explanation:
Citrate chelates ionized Ca²⁺ → hypotension, arrhythmias. Treat with IV calcium gluconate.
12. A patient with confusion and Na⁺ 113 receives 3% NaCl x3 boluses. After 6 hours, Na⁺ = 124. What should be done?
A. Repeat bolus
B. Stop fluids, give DDAVP ✅
C. Switch to normal saline
D. No intervention
Explanation:
Na⁺ rose >10 mEq in 6 hrs — risk of ODS → STOP correction, consider desmopressin to prevent further rise.
13. What is the safest way to replace calcium in a patient with mild hypocalcemia?
A. Calcium chloride bolus
B. Calcium gluconate diluted IV ✅
C. Calcium + phosphate in same IV
D. Push Ca gluconate rapidly in NS
Explanation:
Diluted Ca gluconate over 10–20 min is safest peripheral option.
14. In DKA, which electrolyte must be monitored most closely after starting insulin?
A. Calcium
B. Chloride
C. Phosphate ✅
D. Bicarbonate
Explanation:
Insulin → drives PO₄³⁻ into cells → hypophosphatemia → risk of hemolysis, weakness
15. Which fluid choice is best for correcting metabolic alkalosis with hypochloremia?
A. PlasmaLyte
B. ½ NS
C. Normal Saline ✅
D. Ringer’s lactate
Explanation:
NS = high Cl⁻ content → corrects Cl⁻ deficit and metabolic alkalosis.
1️⃣3️⃣Pocket ICU Guide – Electrolyte Cheat Sheet, Mnemonics & Red Flags
Designed for rapid reference at the bedside, during ward rounds, emergencies, or teaching. This section summarizes all key actions from the guide into one printable page — clinically elegant and focused.
🩺 Target Electrolyte Ranges (ICU Reference)
| Electrolyte | Normal Range | Critical Thresholds |
|---|---|---|
| Na⁺ | 135–145 mEq/L | <120 or >160 |
| K⁺ | 3.5–5.0 mEq/L | <2.5 or >6.0 |
| Ca²⁺ (total) | 8.5–10.5 mg/dL | <7 or >13 |
| Ca²⁺ (ionized) | 1.1–1.3 mmol/L | <0.9 or >1.5 |
| Mg²⁺ | 1.5–2.5 mEq/L | <1.0 or >4.0 |
| Phosphate | 2.5–4.5 mg/dL | <1 or >8 |
| Cl⁻ | 96–106 mEq/L | Variable (acid-base dependent) |
💉 Maximum Safe Infusion Rates (Central vs. Peripheral)
| Electrolyte | Max Peripheral | Max Central | Notes |
|---|---|---|---|
| K⁺ | 10–20 mEq/hr | 40 mEq/hr | ECG monitoring required if >20 |
| Mg²⁺ | 1 g/hr | 2 g/hr | Push only in cardiac arrest |
| Ca gluconate | 1 amp/10–20 min | Multiple amps | Avoid mixing with phosphate |
| Phosphate | 15 mmol/6 hrs | 30 mmol/6 hrs | Avoid rapid shifts |
| 3% NaCl | Not preferred | 100 mL over 10 min | Central line required |
🧮 Deficit Formulas at a Glance
| Electrolyte | Formula |
|---|---|
| K⁺ | 0.3 × weight(kg) × (3.5 − K⁺) |
| Ca (corrected) | Ca + 0.8 × (4 − albumin) |
| Free Water Deficit | TBW × [(Na⁺/140) − 1] |
| PO₄³⁻ | 0.32–0.64 mmol/kg |
| Mg²⁺ | Empirical (2–4 g/day) unless severe |
🧠 Red Flags Mnemonic – “KISS-CaMP”
🔸 K⁺ low but not responding? → Check Mg²⁺
🔸 Insulin → watch K⁺ & PO₄³⁻ drop
🔸 Seizures + low Na⁺ → Use 3% cautiously
🔸 Suction/vomiting → Cl⁻ & K⁺ loss = alkalosis
🔸 Ca²⁺ + Phosphate → never mix (precipitation)
🔸 Magnesium → always replete first
🔸 Phosphate <1 mg/dL → weaning failure, hemolysis
📦 Ampoule Reference
| Drug | Volume | Electrolyte |
|---|---|---|
| KCl 7.5% | 10 mL | 10 mEq |
| KCl 15% | 10 mL | 20 mEq |
| MgSO₄ 50% | 2 mL | 1 g = 8 mEq |
| Ca gluconate 10% | 10 mL | ~90–100 mg elemental |
| NaPO₄ / KPO₄ | 1 mL | 3 mmol phosphate |
| 3% NaCl | 100 mL | 51.3 mEq Na⁺ |
🧬 Replacement Priority – Always Remember:
1️⃣ Magnesium FIRST
2️⃣ Potassium SECOND
3️⃣ Phosphate/Ca as needed
4️⃣ Always correct volume before concentration
🎯 Final Pocket Tip
📍 In any unexplained arrhythmia, ventilator weaning failure, or refractory electrolyte disturbance — check magnesium, phosphate, and calcium together.
🔟 Final Words
Electrolyte management in the ICU is more than just correcting numbers — it’s about preserving cardiac rhythm, preventing respiratory failure, and restoring neuromuscular function. From the silent depletion in refeeding syndrome to the deadly peaks of hyperkalemia, the stakes are high — especially in developing countries and limited-resource environments.
This guide was crafted to deliver a clear, structured, and practical approach to diagnosing, correcting, and monitoring electrolyte imbalances — from potassium to phosphate, from magnesium to chloride. Whether you're managing a transfusion-induced hypocalcemia or titrating phosphate in a malnourished patient, let this guide serve as your clinical compass at the bedside.
Stay focused.
Stay adaptive.
Act with precision.
📌 Prepared for Dr. Amir Fadhel — Specialist in Anesthesiology and Critical Care
Created: 03/06/2025
Last Updated: 03/06/2025
Explore the full collection of completed guides at:
🔗 Mastery Guide Series: https://justpaste.it/jkd89
