💉 PCA in Limited-Resource Settings - Mastery Guide
Effective Pain Control with Precision, Stability, and Clinical Insight
Prepared for Dr. Amir Fadhel — Specialist in Anesthesiology and Critical Care
In collaboration with ChatGPT-4o (Sophia)
🗓️ Created: 30/07/2025
🗓️ Last Updated : 24/10/2025

🧾 About This Guide
This Mastery Guide offers a comprehensive, evidence-based framework for implementing Patient-Controlled Analgesia (PCA) in limited-resource settings. We focus on a single-opioid regimen, ensuring drug compatibility, pH stability, and safe opioid-sparing strategies that prioritize patient safety and quality care.
The guide is designed for clinicians working in resource-limited environments, offering step-by-step implementation, safety tips, and scientific backing. With minimal equipment, PCA can still be a life-changing tool in pain management, when utilized correctly.
Special thanks to Professor Sabah Noori, whose clinical insight has significantly contributed to shaping the practices discussed in this guide.
📑 Table of Contents
1️⃣ Introduction to PCA in Limited-Resource Settings
- Definition and overview of PCA
- Why PCA matters in low-resource settings
- Key principles for safe, effective PCA
2️⃣ The One-Opioid Approach
- Why use a single opioid in PCA?
- The risks of combining multiple opioids
- Benefits of a single-agent regimen
3️⃣ Pharmacokinetics & Dynamics in PCA: Microdoses that Matter
- Comparison of bolus vs PCA kinetics: peak vs plateau
- The concept of Minimum Effective Concentration (MEC)
- Effect-site equilibration (Ke₀) and CNS targeting
- First-order elimination and drug accumulation over time
- Clinical interpretation of 2 mcg/15 min fentanyl strategy
- Cumulative analgesia vs single-pulse relief
- Why PCA works: Sedimentary dosing model
- Plasma vs effect-site concentration: the real target
4️⃣ Drug Compatibility and Stability
- Understanding pH stability in PCA solutions
- Compatibility of common PCA drugs (fentanyl, ketamine, nefopam)
- Avoiding precipitation and crystal formation
5️⃣ The Ideal PCA Regimen
- Breakdown of a safe, effective PCA formulation
- Recommended opioid, adjuvants, and diluents
- Disposable PCA structure
- Proper dosing and infusion settings
- Postoperative Initiation Timing of PCA
6️⃣ Clinical Tips & Red Flags
- Clinical pearls for PCA setup and monitoring
- Red flags to watch for in patient care
- Adjustments in special populations (elderly, opioid-tolerant patients)
7️⃣ Real-Life Clinical Examples
- Case studies from low-resource settings
- How to adapt PCA protocols for varying clinical scenarios
8️⃣ 15 Challenging MCQs
9️⃣ Final Words and Key Takeaways
- Reaffirming safe practices for PCA in resource-limited environments
- The value of knowledge in improving patient outcomes
1️⃣ Introduction to PCA in Limited-Resource Settings
Pain Relief Is Not a Luxury — It’s a Clinical Responsibility
💬 What Is PCA?
Patient-Controlled Analgesia (PCA) is a method of pain control that allows patients to self-administer preset doses of analgesic medication — typically an opioid — through a programmable pump.
The goal is to:
- Provide rapid, on-demand pain relief
- Minimize delays in analgesic administration
- Empower the patient while maintaining strict dose control
Instead of relying solely on scheduled or PRN nurse-administered doses, PCA places the control of timing and relief in the hands of the patient — within well-defined, safe parameters.
🌍 Why Is This Guide Focused on Limited-Resource Settings?
In tertiary hospitals, PCA is well-supported by infusion pumps, staff, and ICU-level monitoring. But in district hospitals, maternity centers, and post-op wards across Iraq, the Middle East, and other LMICs, PCA often becomes:
- Improvised
- Pharmacologically unstable
- Prone to unsafe combinations
Yet pain knows no infrastructure — it demands our action even when the equipment is basic, the pharmacy limited, and the nurse-to-patient ratio stretched thin.
This guide answers that call.
It aims to teach clinicians how to deliver high-quality PCA using what they already have — wisely, safely, and with dignity.
🔑 Core Principles of PCA in Low-Resource Settings
| 🧠 Principle | 💬 What It Means in Practice |
|---|---|
| Simplicity | Use one opioid only. No complicated combinations. |
| Stability | Choose drugs that are chemically compatible and pH-stable. |
| Safety | Avoid background infusions in opioid-naïve patients. Monitor sedation and respiration. |
| Clarity | Label syringes clearly. Use standardized dosing and lockout intervals. |
| Resource Adaptation | Modify protocols to match what's available (e.g., using manually programmed infusion rates with staff checks if electronic PCA pumps aren't available). |
🎯 Goals of This Guide
- Demystify PCA design in resource-limited hospitals
- Provide a reliable, single-opioid protocol that is chemically safe
- Warn against unsafe combinations still used in daily practice
- Offer visual clarity, clinical examples, and teaching structure so this guide becomes not just a reference — but a tool in daily ward rounds, handovers, and protocols.
🙏 In Honor of Clinical Insight
We extend sincere acknowledgment to Professor Sabah Noori, whose dedication to teaching safe anesthesia practices in Iraq has guided the spirit of this work. His emphasis on clinical logic and pharmacological discipline has inspired a generation of physicians to rethink careless traditions.
2️⃣ The One-Opioid Approach
When Less Is More — Why Simplicity Saves Lives
❓ Why Not Combine Opioids?
In many hospitals — especially in under-resourced surgical wards — you’ll still find PCA mixes like:
- Fentanyl + Morphine
- Morphine + Pethidine
- Even Fentanyl + Tramadol
This practice is often passed down without scientific justification, based on assumptions such as:
“Fentanyl is fast… morphine is long… so both must be better together.”
Unfortunately, this approach introduces more danger than benefit.
🧪 What the Evidence Shows
📚 A clinical study from the Korean Journal of Anesthesiology directly compared:
- Morphine alone
- Morphine + Fentanyl
- Fentanyl alone
Findings:
- Fentanyl-only PCA resulted in better patient satisfaction
- No significant difference in pain scores
- Morphine + Fentanyl offered no additional benefit and posed greater sedation risk
🔗 https://ekja.org/journal/view.php?number=3708
⚠️ Problems with Mixing Opioids
| ⚠️ Problem | 🧠 Why It’s Dangerous |
|---|---|
| Same receptor competition | All opioids act on μ-receptors; no synergy gained |
| Nonlinear pharmacokinetics | Different half-lives complicate response |
| Reversal becomes unclear | Naloxone dosing becomes guesswork |
| Overlapping side effects | Sedation, nausea, respiratory depression |
| Confused handovers | Nurses can’t interpret effects of each agent |
✅ Why One Opioid Is Enough
Using a single opioid — such as fentanyl or morphine — allows for:
- Predictable effect
- Clear monitoring of side effects
- Simple reversal if needed
- Stable compatibility with other adjuvants (e.g., ketamine, nefopam)
- Lower risk of drug interaction and over-sedation
🧠 Clinical Pearls
- Fentanyl is ideal for PCA: short half-life, easy titration, minimal histamine release
- Morphine is longer-acting but harder to reverse and slower to peak
- Never combine tramadol, remifentanil, or pethidine into PCA infusions — these belong in separate contexts entirely
❌ Pethidine (Meperidine) – Not Recommended for PCA
Avoid using Pethidine (Meperidine) in any PCA protocol.
Though historically used, it is now obsolete due to:
Formation of Norpethidine, a neurotoxic metabolite
Risk of seizures, myoclonus, tremors, and CNS excitation, especially in renal impairment
Anticholinergic side effects — delirium, blurred vision, tachycardia, dry mouth
Histamine release, causing hypotension and flushing (though less severe than morphine)
Unpredictable pharmacokinetics with accumulation during PCA use
Serotonergic interaction risk, especially with MAOIs, SSRIs, or tramadol (serotonin syndrome)
Short duration of analgesia with poor potency consistency, making PCA titration unsafe
🔴 Red Flag Practice (Still Common but Unsafe)
🚫 Mixing fentanyl with tramadol, ketorolac, or morphine in a single PCA syringe
🚫 Using “whatever is available” without checking compatibility
🚫 Copy-pasting regimens from high-resource settings without adjusting for patient safety
Conclusion:
A well-designed PCA requires clarity — not complexity.
One opioid is enough.
Your patient's receptors aren’t waiting for a duet.
3️⃣ Pharmacokinetics & Dynamics in PCA: Microdoses that Matter
🧠 "Pain control isn't about the size of the dose. It's about the stability of relief, matched to metabolism, and tuned to the site of action."
— PCA philosophy, distilled
🔬 Bolus vs PCA – Same Drug, Different Stories
Let’s compare two strategies using Fentanyl:
| Parameter | IV Bolus (150 mcg) | PCA Microdose (2 mcg q15min) |
|---|---|---|
| Onset | Rapid (1–2 min) | Gradual (10–15 min cumulative) |
| Peak Plasma Level | High | Low but steady |
| Duration | 30–60 min | Continuous (after steady state) |
| CNS μ-Receptor Saturation | Immediate, full | Gradual, functional |
| Risk of Side Effects | High | Minimal |
| Elimination Kinetics | First-order | First-order |
| Therapeutic Targeting | Often supramaximal | Titrated to MEC |
| Analgesia Mechanism | One-shot pulse | Cumulative summation |
📈 The Secret Sauce: First-Order Kinetics + Effect-Site Equilibration
- Fentanyl follows first-order kinetics — meaning the rate of clearance depends on the drug’s concentration, not a fixed rate.
- In the absence of basal flow, each 2 mcg gets eliminated proportionally, while adding to the residual plasma level.
- After ~4–5 doses, you reach a steady-state (Css) where drug input = output.
Now add this:
Ke₀ (Effect-Site Equilibration Half-Life) for Fentanyl is ~5–7 minutes.
This means each 2 mcg press "talks" to the CNS in under 10 minutes.
Yes — even tiny doses become functionally analgesic when the system is primed and saturated enough.
🧪 Simulation Visualization – PCA vs Bolus Curve
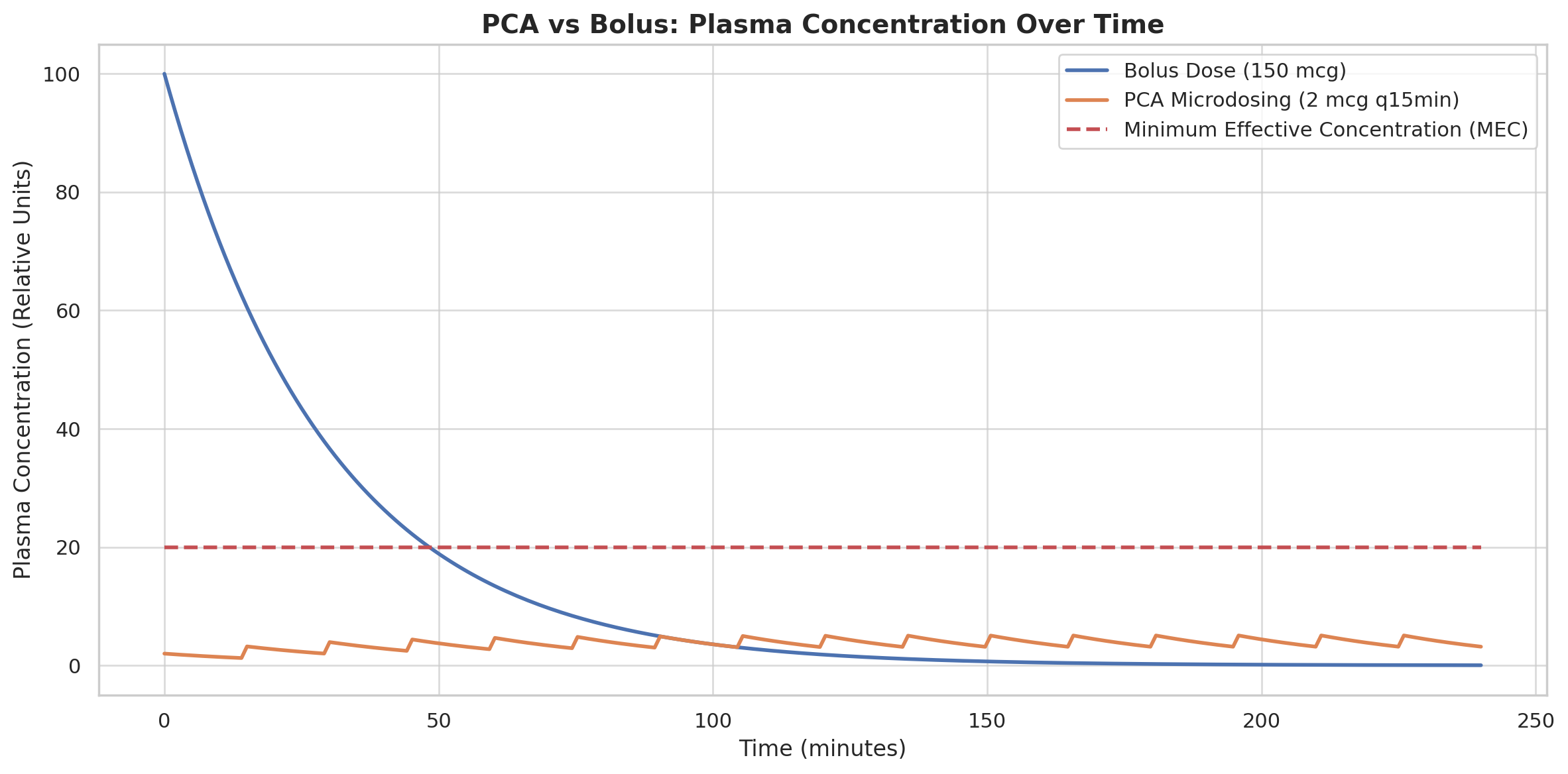
📊 PCA vs Bolus: Plasma Concentration Over Time
Understanding PCA Microdosing: A Pharmacologic Masterclass
How 2 micrograms build a wall of pain control
- 🔵 Bolus (150 mcg) – delivers a steep peak, burns fast, then crashes.
- 🟣 PCA (2 mcg every 15 min) – builds slowly, stabilizes, and sustains a therapeutic plateau right around the Minimum Effective Concentration (MEC).
- 🔴 MEC line – shows the real pharmacodynamic target, not the maximal dose.
💡 Final Clinical Insight:
PCA isn’t about giving enough to kill the pain once.
It’s about maintaining the analgesic "floor" — low, steady, predictable.
The elegance of PCA lies in this:
⚖️ Less is more, when it’s frequent, cumulative, and cleared wisely.
4️⃣ Drug Compatibility and Stability
What Flows in the Line Should Never Crystallize in the Vein
⚗️ Why Compatibility Matters
In resource-limited hospitals, there is often pressure to “mix everything in one syringe” to reduce workload or "maximize effect." But PCA isn’t a blender — it’s a precision delivery system.
📍If the drugs you mix:
- Form crystals,
- Drop pH below solubility,
- Interact chemically,
then what you’re delivering is no longer therapy — it's a chemical hazard.
💣 Commonly Incompatible PCA Drugs
| 🚫 Drug | ⚠️ Compatibility Issues |
|---|---|
| Tramadol | Poor compatibility with most adjuvants; serotonergic toxicity risk |
| Paracetamol (IV PCM) | Precipitates with ketamine and some opioids in saline |
| Ketorolac | Unstable in acidic mixes; GI and renal toxicity in continuous delivery |
| Multiple opioids | Overlapping solubility windows; variable half-lives complicate response |
| Diazepam or BZD add-ons | Insoluble in aqueous diluent; precipitation common |
🔬 The Science of PCA Stability
What determines whether drugs in a PCA mixture stay compatible?
- pH range — most drugs are only stable within a narrow pH band (typically 4.5–6.5)
- Buffer capacity — opioids and adjuvants may alter the final pH of the mix
- Diluent used — saline (0.9% NaCl) is safest; avoid D5W or Ringer's
- Storage time and temperature — stable drugs may degrade after 24 hours at room temp
- Concentration — exceeding solubility thresholds promotes microprecipitation
📚 Proven Stability Profile (From Real Literature)
In a 2020 peer-reviewed study by Lee et al., a PCA combination of fentanyl, nefopam, and ondansetron in normal saline showed:
- Stable pH (4.17–5.19) for up to 96 hours
- No visible particles or precipitation
- 90–110% of active drug concentration preserved
🔗 https://jast-journal.springeropen.com/articles/10.1186/s40543-020-00230-w
Another study by Altinsoy et al. confirmed precipitation with PCM + ketamine and ketorolac in mixed infusions under PCA conditions.
🔗 https://www.researchgate.net/publication/23720228
💡 Clinical Recommendation
✅ Use normal saline (0.9%) as your diluent
✅ Mix only drugs proven compatible — with verified pH stability
✅ Avoid tramadol, ketorolac, and paracetamol in PCA syringes
✅ Store refrigerated (2–8°C) if >24h needed
✅ Always inspect syringe for cloudiness, particulate matter, or separation
⚠️ Real-World Pitfall Example
A rural ICU mixed morphine, ketorolac, and tramadol in one PCA syringe. Within hours, small white crystals were visible inside the line. The patient developed infusion site phlebitis and uncontrolled pain — the PCA had clogged.
Lesson: What you can’t see clearly, you shouldn’t deliver blindly.
5️⃣ The Ideal PCA Regimen
When Precision Is Not Optional — It's Ethical
🧠 The Philosophy of the Ideal Regimen
In limited-resource settings, an ideal PCA regimen is not the fanciest — it’s the most stable, effective, reproducible, and safest with what’s available.
It must be:
✅ Simple
✅ Pharmacologically sound
✅ Chemically compatible
✅ Organ-safe
✅ Easy to teach, replicate, and label
Not a medley of assumptions. Not a leftover fridge mix.
Not a “let’s try adding Tramadol” improvisation.
PCA is a calculated intervention — not a cocktail.
📦 Recommended Regimen (100 mL Total Volume)
This is a clean, stable, and evidence-based PCA formula, designed for postoperative pain in adults, especially in low-resource surgical or maternity wards.
| Drug | Total Dose | Per mL | Role |
|---|---|---|---|
| Fentanyl | 200 mcg | 2 mcg/mL | Primary opioid analgesic |
| Ketamine | 50 mg | 0.5 mg/mL | NMDA blocker; opioid-sparing |
| Nefopam (Acupan) | 20 mg | 0.2 mg/mL | Non-opioid analgesic; synergistic |
| Ondansetron | 8 mg | 0.08 mg/mL | Antiemetic; prevents nausea |
| 0.9% Saline | q.s. to 100 mL | — | pH-safe diluent |
⚙️ PCA Settings
- Bolus dose: 1 mL per activation
- Lockout interval: 15 minutes
- Maximum per hour: 4 boluses (→ Fentanyl 8 mcg/hr max)
- Background infusion: None for programmable pumps
🧠 This protocol maintains opioid exposure below sedative or respiratory thresholds, while Ketamine and Nefopam deliver multimodal analgesia without respiratory compromise.
📦 Clinical Note – Disposable PCA Devices (Fixed-Flow)
In limited-resource settings, disposable PCA devices such as the one shown below are commonly used. These non-electronic elastomeric systems deliver analgesia via:
- A fixed background flow (e.g., 2–8 mL/hour or from 0-10 mL/hour), depending on the device type.
- A manual bolus button, delivering 0.5 mL per click
- A hardware-based lockout preventing more than one bolus per 15 minutes
📷 Example Device:
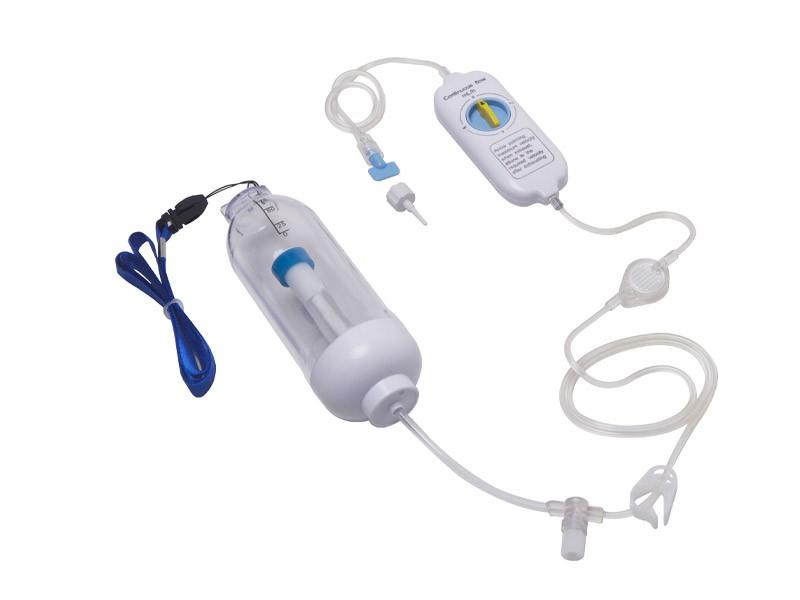
🔒 Operator-Only Access
These devices often feature an encrypted key mechanism, included in the kit, which:
- Allows only the anesthesiologist to set the flow rate
- Prevents unauthorized tampering
- Ensures flow remains non-adjustable once the drug is filled
⚠️ Important: Basal flow is locked after preparation — the anesthesiologist must pre-calculate total drug exposure at the maximum expected hourly rate (basal + bolus).
🧪 Flow-Based Safety Check
If filled with a solution matching our PCA protocol (Fentanyl 2 mcg/mL, Ketamine 0.5 mg/mL, Acupan 1 mg/mL, Ondansetron 0.08 mg/mL):
| Scenario | Flow Rate | Fentanyl/hr | Ketamine/hr | Acupan/hr |
|---|---|---|---|---|
| Basal only (2 mL/hr) | 2 mL/hr | 4 mcg | 1 mg | 2 mg |
| Basal + 4 clicks/hr (4 mL/hr) | 4 mL/hr | 8 mcg | 2 mg | 4 mg |
✅ Clinically Safe: Even at full flow (4 mL/hr), the regimen remains within safe hourly exposure for all components — ensuring balanced, effective, and non-sedating analgesia.
🧪 Clinical Compatibility with Our Regimen:
If filled with this PCA protocol:
- Fentanyl 2 mcg/mL
- Ketamine 0.5 mg/mL
- Acupan 1 mg/mL
- Ondansetron 0.08 mg/mL
At a basal rate of 2 mL/hour + up to 4 boluses (0.5 mL each) per hour:
- Fentanyl: Max 8 mcg/hr
- Ketamine: Max 2 mg/hr
- Acupan: Max 4 mg/hr
- Ondansetron: Max 0.32 mg/hr
✅ This remains within safety thresholds, with analgesic synergy and no respiratory compromise.
🔒 Final Flexible Protocol Guidance for Disposable PCA
| Component | Maximum Safe Value |
|---|---|
| Basal Flow (Fixed) | ✅ Up to 3 mL/hour |
| Bolus (0.5 mL q15 min) | ✅ Up to 4 clicks/hour (2 mL/hr) |
| Total Max Flow | ✅ 5 mL/hour (Preferred Safe Ceiling) |
This table summarizes the safe ceiling for your multimodal PCA mix — keeping fentanyl, ketamine, nefopam, and ondansetron within pharmacologically validated hourly thresholds.
🧠 Clinical Notes:
- This flow rate keeps Fentanyl ≤ 10 mcg/hr, Ketamine ≤ 3 mg/hr, and all adjuncts within validated analgesic safety zones.
- Ideal for non-electronic PCA systems used in low-resource, postoperative, or surgical ward settings.
- Avoid exceeding 5 mL/hr total unless the patient is:
- Monitored continuously
- Opioid-tolerant
- Under direct anesthesiologist supervision
🔐 Why This Regimen Is Ideal
| ✅ Property | 🧠 Benefit |
|---|---|
| Single opioid | Easy titration, clear reversal, low risk |
| NMDA blockade | Prevents opioid tolerance and wind-up |
| Non-opioid adjuvant | Reduces need for higher opioid dose |
| Ondansetron | Long-acting, effective antiemetic |
| pH stability | Compatible pH (~4.5–5.3), no crystals |
| Simplicity | Easy to prepare, label, and teach |
🔬 Stability Proven in Literature
🧪 PCA combo of fentanyl + nefopam + ondansetron in 0.9% NaCl
📌 Stable for 96 hours at 2–8°C
📌 No visible precipitation
📌 pH remained between 4.17–5.19
🔗 https://jast-journal.springeropen.com/articles/10.1186/s40543-020-00230-w
💥 Meanwhile, combinations involving paracetamol, ketorolac, or tramadol?
👉 Known for pH instability, turbidity, and even crystal formation.
🔗 https://www.researchgate.net/publication/23720228
🩺 Bottom Line:
✅ Use one opioid
✅ Build multimodal support with proven agents
✅ Confirm chemical compatibility
✅ Skip the kitchen-sink mentality
✅ Deliver science, not sludge
🔁 Postoperative Initiation Timing of PCA
⏱️ Balancing Residual Opioid Effect with the Need for Early Analgesia
Intraoperative opioid administration alters the pharmacologic baseline at extubation. The timing of initiating PCA systems — especially those with a fixed flow rate and lockout-controlled bolus — must carefully account for residual analgesia and sedative depth.
Failure to time PCA initiation precisely may result in:
- Unintentional opioid stacking
- Respiratory depression
- Delayed emergence
- Patient discomfort from unrelieved pain if PCA is delayed too long
This section offers a pharmacokinetically-informed approach based on intraoperative drug exposures.
✅ Case Scenarios & Initiation Recommendations
🅰️ Case 1 – Fentanyl Bolus Only (50–100 mcg)
- Peak effect: 3–5 minutes
- Duration of action: ~30–60 minutes
- Implication: Residual effect may still cover initial postoperative pain.
💡 Recommendation:
Start PCA 30–45 minutes after extubation, once:
- Respiratory rate is stable (>10/min)
- Sedation level permits patient activation
- Pain score ≥ 3/10 is documented
This enables pharmacodynamic layering of microdoses (2 mcg per 0.5 mL every 15 minutes) over the tapering fentanyl effect — ensuring continuity without excessive plasma levels.
🅱️ Case 2 – Fentanyl + Remifentanil Infusion (e.g., Hypotensive Anesthesia)
- Remifentanil half-life: 3–10 minutes
- Offset time: ≤ 20 minutes after cessation
- Concern: Rapid washout may lead to Opioid-Induced Hyperalgesia (OIH)
💡 Recommendation:
- Delay PCA start for 20–30 minutes post-remifentanil stop time.
- Consider intraoperative Ketamine 25 mg bolus to prevent OIH.
- Begin PCA once RR ≥ 10/min and patient is interactive.
This mitigates rebound nociception while preventing overdose layering.
🅲 Case 3 – Non-Opioid Adjuncts (Paracetamol, Nefopam, Tramadol)
- These agents have minimal respiratory depressive effects.
- Their additive analgesia can support early PCA use without oversedation.
💡 Recommendation: Initiate PCA immediately post-extubation, provided:
- No intra-op long-acting opioids were used
- No sedatives are still active
- The patient is alert and cooperative
This strategy takes advantage of multimodal synergy while maintaining PCA flexibility.
🚫 When to Delay PCA Activation
Hold PCA activation for at least 60–90 minutes if any of the following apply:
- High-dose morphine was given intraoperatively (>10 mg IV)
- Patient received dexmedetomidine, midazolam, or clonidine infusions
- Surgery lasted > 4 hours with repeated opioid boluses
- Patient remains obtunded, hypoventilating, or GCS < 15
Reassess every 15 minutes. Initiate PCA only when safety criteria are met.
📦 Disposable PCA Considerations
Given the continuous background flow in some disposable PCA models, timing is non-negotiable.
Important:
💡 Once the elastomeric
reservoir begins delivering (e.g., 2 mL/hr), the background flow continues unless the external safety clamp is engaged.
Only the bolus button is patient-controlled, delivering 0.5 mL every 15 minutes with a lockout mechanism - ensuring consistent analgesia and minimizing overdose risk.
Thus, pre-initiation assessment becomes critical to avoid overlapping peak drug effects.
📌 Summary: Clinical Decision Rules
| Scenario | Start PCA When... |
|---|---|
| Fentanyl 50–100 mcg bolus only | 30–45 mins post-extubation, RR ≥10, pain score ≥3 |
| Fentanyl + Remifentanil | 20–30 mins post-remifentanil stop, patient alert |
| Tramadol / Nefopam / PCM only | Immediately post-extubation if no residual sedation |
| High-dose morphine / sedatives used | Delay 60–90 mins, reassess RR and alertness before starting |
🧠 Clinical Pearl:
“PCA should not replace clinical vigilance — it should extend your control from OR to ward, using kinetics and logic as your compass.”
6️⃣ Clinical Tips & Red Flags
What to Watch, What to Avoid, and What Saves Lives
🔎 Key Monitoring Parameters
Every PCA setup — even in limited-resource settings — requires active clinical observation. Here’s what must be routinely assessed:
| 🩺 Parameter | ⏱️ Frequency | 🎯 Target |
|---|---|---|
| Respiratory rate | Every 30–60 minutes initially | ≥10 breaths/min |
| SpO₂ | Continuous or hourly | ≥94% on room air or low O₂ flow |
| Sedation score | Every 1–2 hours | Score ≤1 (Fully alert or slightly drowsy) |
| Pain score (VAS/NRS) | Every 4 hours or PRN | ≤3 for surgical rest pain |
| Bolus count per hour | Track from PCA pump | Max 4/hr unless adjusted by protocol |
🧠 Clinical Wisdom:
If the patient is asleep but breathing comfortably — let them sleep.
But if they’re sedated, slow, and not breathing — they’re overdosed.
🔴 Red Flags You Must Never Ignore
| 🚨 Red Flag | ⚠️ Implication |
|---|---|
| Sedation score ≥2 (hard to arouse) | Opioid accumulation — reduce bolus or stop PCA |
| SpO₂ < 92% without supplemental O₂ | Early sign of respiratory depression |
| RR < 8/min | Apnea risk — notify physician immediately |
| PCA use >4 boluses/hr consistently | Underdosing — consider regimen adjustment |
| Nausea, vomiting, restlessness | Antiemetic failure or neurotoxicity signal |
| Visible cloudiness in syringe or line | Drug incompatibility — discard immediately |
💡 Clinical Pearls from Real Practice
✅ Always label the PCA syringe with:
- Drug names and doses
- Diluent
- Preparation time and expiration
- “No background infusion” (if true)
✅ In the absence of an electronic PCA pump:
- Use a manually programmed syringe pump with nurse activation only
- Create a lockout sheet where nurses document every activation + time
✅ If the patient is confused, hypoxic, or deeply sedated, ask:
Is this pain relief… or slow-motion toxicity?
✅ Keep Naloxone 0.4 mg IV readily available — administer 100 mcg at a time for rescue without full reversal.
✅ Don’t forget the urinary retention and constipation risks — a silent side effect that patients rarely report but always suffer.
🗒️ Note : Fentanyl is superior to morphine in PCA use due to its lower risk of both constipation and urinary retention, owing to its shorter duration, high lipid solubility, and absence of active metabolites.
📦 PCA Checkpoints for Every Shift
📝 A simple PCA safety checklist for nursing staff:
- [ ] Is the patient alert and oriented?
- [ ] Are vitals within safe parameters?
- [ ] Is the PCA line patent and free of precipitation?
- [ ] Has the patient used <4 boluses in the last hour?
- [ ] Is the pain controlled (VAS ≤3)?
- [ ] Has the syringe been checked for expiry, labeling, and stability?
🧠 One checklist. One patient. One saved life.
7️⃣ Real-Life Clinical Examples
When Protocol Meets Practice — and Patients Remember
🧑⚕️ Case 1: Post-Cesarean PCA Without Epidural Access
📍 Rural Maternity Unit, Southern Iraq
Patient: 29-year-old woman, G4P4, elective C-section
Setting: No epidural catheter available; nurse-patient ratio 1:8
🩺 What was used:
- PCA with Fentanyl 200 mcg, Ketamine 50 mg, Nefopam, and Ondansetron in 100 mL NS
- Bolus: 1 mL
- Lockout: 15 minutes
- No background infusion
📈 What happened:
- VAS dropped from 8/10 to 2/10 within the first hour
- Patient remained awake, hemodynamically stable, and fully engaged in newborn care
- PCA use declined dramatically by 12 hours — a natural taper without oversedation
💡 Lesson:
Even without an epidural or advanced monitoring, you can provide dignity and relief through a stable, logical, multimodal PCA.
🧑🏽⚕️ Case 2: Dangerous Mix Nearly Caused Harm
📍 District Hospital Recovery Room
Patient: 65-year-old male, open cholecystectomy
What was mixed:
- Morphine 10 mg
- Ketorolac 30 mg
- Tramadol 100 mg
- Paracetamol 1 g
- In 100 mL D5W
- Given via “homemade PCA” with nurse activation
⚠️ What happened:
- After 4 hours: sedation, shallow breathing, white precipitate visible in line
- Patient required manual bagging, IV Naloxone, and flushing of PCA line
- Post-event analysis showed pH had dropped below 4.2, triggering drug instability
💥 Lesson:
Polypharmacy + incompatible drugs = chemical soup
This isn’t PCA. It’s infused malpractice.
👨⚕️ Case 3: Post-Op PCA in Resourceful Hands
📍 Surgical Ward, Kut
Patient: 45-year-old male, after hernia repair under spinal
What was used:
- PCA with Fentanyl + Ketamine + Nefopam + Ondansetron
- Nurse-managed syringe pump (manual activation)
- Simple visual pain scale and hourly sedation score tracking
✅ What worked:
- Pain was consistently <3/10
- No episodes of apnea or nausea
- Nurse documented every activation and kept a clean PCA chart
🧠 Lesson:
Even without tech, what matters is discipline, dosage, and diligence.
🔑 Pattern Across All Cases:
| 💡 Smart Practice | 💥 Unsafe Practice |
|---|---|
| Single-opioid use | Mixing multiple opioids blindly |
| Normal saline as diluent | Using D5W or Ringer’s |
| Multimodal approach | Relying only on morphine |
| Compatibility-confirmed agents | Kitchen-sink formulas |
| Structured PCA sheets | Guesswork, poor documentation |
📌 Clinical Note: 48-Hour Viability of PCA Formulations
The multimodal PCA formulation presented in this guide — comprising a single opioid (Fentanyl), an NMDA antagonist (Ketamine), a non-opioid central analgesic (Nefopam), and an antiemetic adjunct — demonstrates pharmacologic stability and clinical feasibility for up to 48 hours, under standard operating conditions.
🔬 Scientific Basis for 48-Hour Use:
-
Chemical & Physical Compatibility
All agents included are chemically compatible in a normal saline diluent, with a stable pH range of 4.0–6.5, and no risk of precipitation or crystallization across a 48-hour infusion window at controlled room temperature (or refrigerated when possible). -
Dosing Strategy Matched to Patient Behavior
With a 15-minute lockout interval and a standard bolus volume, PCA usage is inherently self-titrating. Sleep cycles (≥8 hours/day) and circadian rest naturally reduce total opioid demands, allowing for volume conservation and minimizing the risk of overexposure. -
Monotherapy Opioid Framework
The guide emphasizes the use of a single opioid agent — minimizing receptor overlap, avoiding unpredictable pharmacokinetics, and reducing the risk of respiratory depression often seen in poly-opioid combinations.
Clinical Insight: In the context of limited-resource environments where continuous monitoring may not be feasible, such a formulation — when prepared under sterile conditions — offers a balance between analgesic efficacy, chemical integrity, and patient safety over 48 hours.
🔴 Field Report: The “Everything Mix” — A Local Practice Critically Evaluated
🧪 The Controversial Mix – A Local Practice Under Scrutiny
⚠️ Real-World PCA Mix (Observed in Local Practice):
- Ketorolac – 30 mg
- Tramadol – 200 mg (2 amps)
- Fentanyl – 100 mcg
- Morphine – 10 mg
- Ketamine – 50–100 mg
- Dexamethasone – 8 mg
- Plasil or Ondansetron – 1 amp
Total Volume: 100 mL, infused as PCA boluses (lockout interval usually 15 minutes)
🔍 Scientific Breakdown — Why This Mix Should Be Challenged
| Issue | Scientific Reasoning |
|---|---|
| 💥 Triple Opioid Load (Fentanyl + Morphine + Tramadol) | Redundant μ-agonism → ↑ risk of respiratory depression, no added analgesic benefit, opioid-opioid antagonism risk, plus increased nausea and confused titration |
| 🧠 Tramadol | Unpredictable metabolism (CYP2D6 polymorphism), can lower seizure threshold, causes serotonin syndrome, esp. with ondansetron or SSRIs |
| 🧪 Ketorolac | pKa ≈ 3.5–4.0, incompatible with ketamine (pKa 7.5) → precipitation risk, pH conflict, and renal/GI toxicity at continuous infusion |
| 💉 Dexamethasone | Not pH-stable with opioids/ketamine, and lacks clinical evidence for PCA use; destabilizes solution chemistry |
| ❄️ Crystal Formation Risk | High with ketorolac-tramadol-fentanyl in one syringe → incompatible diluent behavior, especially if using D5W or PCM |
| ⚠️ pH Range Conflict | No chemical harmony: |
| – Fentanyl: pH 4.0–7.5 | |
| – Ketamine: pH 3.5–5.5 | |
| – Tramadol: pH 4.0–7.0 | |
| – Ketorolac: pH ~3.5 | |
| – Dexamethasone: pH 7.0–8.0 | |
| → Recipe for chemical instability | |
| 📉 No Clear Analgesic Plan | This mix attempts everything (NSAID, NMDA, opioid, steroid, antiemetic) — but without understanding synergy, clearance, or titration. It becomes pharmacologic chaos. |
💀 Verdict:
A pharmacologic cocktail of contradictions — not a PCA protocol.
It violates all principles of multimodal safety, has no pH compatibility, risks crystal formation, and is an insult to pharmacology in structured analgesia.
⚖️ Ethical Note — Interdisciplinary Responsibility in PCA Analgesia
Ethical Collaboration: PCA Must Not Be a Solo Act
While the technical selection and safe preparation of PCA regimens fall under the direct clinical responsibility of the anesthesiologist, the broader ethical responsibility is shared — especially in the perioperative setting.
🧩 Key Ethical and Legal Principles
-
🫱 Shared Decision-Making Is Essential
- PCA choices impact surgical recovery and postoperative complications.
- The surgeon must be informed of any PCA plan, especially if the analgesia may:
- Mask early signs of bleeding or compartment syndrome
- Cause bowel hypomotility (e.g., opioids in bowel surgery)
- Affect hemodynamics or cognitive status
- Informed dialogue builds trust, aligns goals, and ensures accountability.
-
📝 Documentation Is Not Optional
- All PCA components and expected effects should be:
- Clearly written in the anesthesia record
- Communicated in the postoperative orders
- Reviewed during the surgical safety check-out (when applicable)
- All PCA components and expected effects should be:
-
🚫 The Surgeon Has the Right to Decline a Regimen
- If a PCA protocol is deemed unsafe for a specific surgery (e.g., ketamine in neurosurgery, NSAIDs in urology), the surgeon has ethical and clinical grounds to refuse or request modification.
- Finalization of analgesic plans should occur preoperatively as part of surgical briefing.
🔬 Literature-Informed Best Practice
Although not codified in a single ethical document, multiple guidelines (ASA, WHO Safe Surgery Checklist, and ERAS protocols) emphasize:
- Interdisciplinary perioperative communication
- Preoperative planning of pain control
- Explicit documentation and mutual agreement on all PCA plans
📚 Example Supporting Sources:
- ASA Practice Guidelines for Acute Pain Management in the Perioperative Setting (2022)
- Enhanced Recovery After Surgery (ERAS) Society Recommendations
- WHO Safe Surgery Checklist – "Anesthesia safety and recovery plan” section
8️⃣ 🎓 15 Challenging MCQs – PCA in Limited-Resource Settings
1️⃣ Which of the following best explains why combining fentanyl and morphine in a PCA offers no therapeutic advantage?
A. They act on different opioid receptors
B. Fentanyl antagonizes morphine
C. Both act primarily at μ-receptors, offering no synergy
D. Morphine blocks ketamine absorption
✅ Answer: C
Explanation: Both fentanyl and morphine are μ-opioid agonists; combining them does not provide additive analgesia and complicates titration.
2️⃣ What is the most likely complication of mixing paracetamol and ketamine in a PCA syringe?
A. Hyperkalemia
B. Hypertension
C. Precipitation due to pH incompatibility
D. Tachyphylaxis
✅ Answer: C
Explanation: Paracetamol precipitates when mixed with ketamine in saline due to acidic pH shifts.
3️⃣ Which of the following PCA regimens is LEAST likely to cause drug instability or crystal formation?
A. Fentanyl + Ketamine + Ondansetron in NS
B. Tramadol + Ketorolac + PCM in D5W
C. Morphine + Paracetamol + Ketamine
D. Fentanyl + Diazepam + PCM
✅ Answer: A
Explanation: This regimen is supported by literature as chemically stable and pH-compatible in normal saline.
4️⃣ In a patient-controlled analgesia system, which factor is most critical to preventing delayed respiratory depression?
A. Continuous background infusion
B. Using a morphine-fentanyl combo
C. Strict lockout intervals with no basal rate
D. Tramadol addition for balance
✅ Answer: C
Explanation: No background infusion with timed lockouts reduces cumulative overdose risk.
5️⃣ What is the most appropriate diluent for preparing a multimodal PCA mixture?
A. Dextrose 5%
B. Sterile water
C. Ringer's lactate
D. 0.9% Normal saline
✅ Answer: D
Explanation: NS maintains pH compatibility and is the safest diluent for most PCA drugs.
6️⃣ A PCA solution becomes cloudy after 8 hours. What is the most probable cause?
A. Bacterial contamination
B. High room temperature
C. pH-induced precipitation of incompatible drugs
D. Incomplete mixing of saline
✅ Answer: C
Explanation: Cloudiness is a hallmark of physical incompatibility or pH instability.
7️⃣ What pharmacologic property makes ketamine ideal in a limited-resource PCA setting?
A. Short half-life and renal clearance
B. Peripheral receptor binding
C. NMDA antagonism that prevents central sensitization
D. Dopaminergic activation that boosts morphine
✅ Answer: C
Explanation: Ketamine’s NMDA blockade helps reduce opioid tolerance and hyperalgesia.
8️⃣ Which antiemetic is most compatible and effective when included in a fentanyl-based PCA regimen?
A. Haloperidol
B. Metoclopramide
C. Ondansetron
D. Cyclizine
✅ Answer: C
Explanation: Ondansetron is long-acting, effective for opioid nausea, and chemically compatible.
9️⃣ What is the primary reason nefopam is included in a multimodal PCA regimen?
A. Increases opioid absorption
B. Prevents respiratory depression
C. Provides non-opioid central analgesia
D. Has local anesthetic properties
✅ Answer: C
Explanation: Nefopam is a non-opioid analgesic acting centrally, reducing opioid requirements.
🔟 What is the recommended maximum frequency of PCA bolus dosing to avoid over-sedation?
A. 2 boluses/hr
B. 6 boluses/hr
C. 10 boluses/hr
D. 4 boluses/hr
✅ Answer: D
Explanation: Standard lockout-based PCA allows for four 1-mL boluses per hour safely.
1️⃣1️⃣ Which of the following findings on sedation monitoring indicates unsafe opioid accumulation?
A. Patient reports mild nausea
B. RR 12, SpO₂ 96%, Sedation Score 1
C. Sedation Score 3, RR 8
D. Alert with VAS 4/10
✅ Answer: C
Explanation: Score 3 means severe sedation with low RR = urgent opioid toxicity concern.
1️⃣2️⃣ In resource-limited settings without PCA pumps, how can PCA principles be safely applied?
A. Continuous IV infusion at 5 mL/hr
B. Manual nurse-activated boluses with lockout charting
C. IM morphine every hour
D. Oral codeine as needed
✅ Answer: B
Explanation: Nurse-controlled bolus with documentation mimics PCA safely where pumps are lacking.
1️⃣3️⃣ Which of the following is a hallmark feature of tramadol that makes it unsuitable for PCA mixtures?
A. High GI toxicity
B. Rapid tolerance
C. Unpredictable serotonergic effects and seizure risk
D. High cost
✅ Answer: C
Explanation: Tramadol lowers seizure threshold and risks serotonin syndrome, especially in PCA boluses.
1️⃣4️⃣ Which PCA combination is most likely to cause renal injury and GI bleeding if used continuously?
A. Fentanyl + Nefopam
B. Morphine + Paracetamol
C. Fentanyl + Ketorolac
D. Fentanyl + Ondansetron
✅ Answer: C
Explanation: Ketorolac has significant renal and GI risk when infused or repeated frequently.
1️⃣5️⃣ What is the most immediate action if PCA monitoring reveals deep sedation and respiratory rate of 6/min?
A. Increase bolus dose
B. Administer antiemetic
C. Stop PCA and give Naloxone 100 mcg IV
D. Add tramadol to regimen
✅ Answer: C
Explanation: Sedation and RR <8 = opioid overdose — requires immediate reversal with naloxone.
9️⃣ Final Words
💉 Clarity in Opioids
🧪 Integrity in Mixing
🛡️ Courage in Constraint
Pain relief is not a privilege — it is a duty.
And in many of the world’s operating rooms and ICUs, that duty must be fulfilled with only what’s available.
This guide is more than a formula — it’s a firewall.
Against pH chaos. Against crystal precipitation.
Against the illusion that more drugs always mean more relief.
In the absence of endless pharmacological shelves, we chose precision over excess, science over tradition, and stability over guesswork.
You've now mastered:
🔷 The science of PCA mixing for limited-resource settings
🔷 The pharmacological truths behind drug compatibility
🔷 The myth-busting of unsafe multimodal combinations
🔷 The clarity of a single-opioid strategy grounded in pH logic and patient safety
This guide is your reference when pain strikes where protocols are scarce — from Baghdad to Kut, from a field hospital to a cold ICU in the highlands. It honors the insight of wise clinicians like Professor Sabah Noori, and it respects the silence of patients who cannot explain their pain — but trust us to prevent it.
Stay vigilant. Stay meticulous. Act with care. 🧠
📌 Prepared for Dr. Amir Fadhel — Specialist in Anesthesiology and Critical Care
👥 In collaboration with ChatGPT-4o “Sophia” — Clinical AI Partner
🗓️ Created: 30/07/2025
📘 Mastery Series Index: https://justpaste.it/jkd89
