🌬️ ARDS Mastery Guide
Prepared for Dr. Amir Fadhel — Specialist in Anesthesiology and Critical Care
Powered by ChatGPT-4o | Clinical Teaching & ICU Reference
🤝 About This Guide
This guide is the result of a unique collaboration between Dr. Amir Fadhel, anesthesiologist and critical care specialist, and Sophia, a cutting-edge AI assistant powered by OpenAI’s ChatGPT-4o model—one of the most advanced tools for clinical reasoning and medical education available today.
Together, we present a meticulously crafted clinical journey into one of the most complex and life-threatening conditions in critical care:
📘 Acute Respiratory Distress Syndrome (ARDS)
This guide has been shaped not only by AI-supported evidence synthesis and visual design—but also by the academic inspiration drawn from the lecture of Assist. Prof. Dr. Waleed Al-Ansari / Critical Care Medicine, whose teaching and pathophysiologic insights are respectfully integrated throughout this work.
This comprehensive guide is part of a growing series that includes:
🔹 Arterial Blood Gas (ABG) Interpretation
🔹 Mechanical Ventilation: Modes, Basics, and Waveforms
With this new ARDS Mastery Guide, we take a deep dive into:
-
🔸 Updated definitions (Berlin, PALICC)
-
🔸 Detailed pathophysiology across exudative, proliferative, and fibrotic phases
-
🔸 Differentiation from mimics like cardiogenic edema
-
🔸 Modern ICU management including prone positioning, ECMO, and LPV
-
🔸 Adapted strategies for resource-limited settings
-
🔸 Special populations including pediatrics, pregnancy, and trauma
-
🔸 Clinical red flags, pearls, and real ICU scenarios
-
🔸 15 Expert-level MCQs to test understanding
Whether you are a medical student, anesthesia technician, resident, or ICU clinician, this guide brings you visual clarity, clinical structure, and global applicability—making it your go-to reference for mastering ARDS.
🔷 For subscription-based AI clinical tools, educational collaboration, or to contribute content, reach out to Dr. Amir Fadhel, or explore OpenAI’s professional services.
1️⃣ Introduction to ARDS
🩺 What is ARDS? – A Unified, Modern Definition
Acute Respiratory Distress Syndrome (ARDS) is a clinically defined, life-threatening form of acute, non-cardiogenic respiratory failure that occurs in response to a wide variety of direct and indirect lung injuries.
It is characterized by:
-
Acute onset (within 1 week of a known clinical insult)
-
Bilateral pulmonary infiltrates on chest imaging (not fully explained by effusion, collapse, or nodules)
-
Hypoxemia, defined by a PaO₂/FiO₂ ratio ≤300 mmHg while on PEEP ≥5 cmH₂O, and
-
Exclusion of cardiac failure or fluid overload as the primary cause of pulmonary edema
(Berlin Definition, 2012)
🔬 Pathophysiologic Insights
ARDS is underpinned by diffuse alveolar-capillary membrane injury, driven by:
-
Neutrophil recruitment and activation through ICAM-1 and ELAM-1
-
Loss of surfactant, alveolar flooding, and formation of hyaline membranes
-
Increased capillary permeability leading to protein-rich pulmonary edema
-
Ineffective hypoxic pulmonary vasoconstriction (HPV), resulting in right-to-left shunting
-
Reduced lung compliance, increasing the work of breathing and leading to refractory hypoxemia
📌 In summary, ARDS represents the final common pathway of acute alveolar injury, where structural damage, dysregulated inflammation, and impaired gas exchange culminate in respiratory failure that demands early recognition and targeted supportive care.
🧾 The Most Updated Definition: Berlin Definition (2012)
As of 2024, the Berlin Definition remains the globally recognized standard for defining ARDS:
| 🔍 Component | ✅ Berlin Criteria (2012) |
|---|---|
| 1️⃣ Timing | Within 1 week of a known clinical insult OR new/worsening respiratory symptoms |
| 2️⃣ Imaging | Bilateral opacities on chest X-ray or CT—not fully explained by effusion, collapse, or nodules |
| 3️⃣ Origin of Edema | Respiratory failure not fully explained by cardiac failure or fluid overload |
| 4️⃣ Oxygenation (on PEEP ≥5 cmH₂O) | |
| ➤ Mild ARDS | PaO₂/FiO₂ = 200–300 mmHg |
| ➤ Moderate ARDS | PaO₂/FiO₂ = 100–200 mmHg |
| ➤ Severe ARDS | PaO₂/FiO₂ ≤100 mmHg |
📌 Note: PaO₂/FiO₂ ratio must be measured under positive pressure ventilation with PEEP ≥5 cmH₂O.
🔄 Are There Any Newer Updates (2024)?
While the Berlin Definition remains standard, discussions are ongoing to:
-
Include ARDS in non-intubated patients (e.g., HFNC, CPAP users)
-
Use lung ultrasound as a diagnostic tool
-
Reassess PEEP and FiO₂ thresholds in light of newer oxygenation strategies
However, no official update or replacement has been universally adopted yet.
📜 Historical Context
-
🗓️ 1967: ARDS first described by Dr. Ashbaugh et al. as a condition with dyspnea, cyanosis, bilateral infiltrates, and poor response to oxygen.
-
🧒 Initially called “Adult” Respiratory Distress Syndrome to distinguish it from neonatal RDS.
-
🔁 Name evolved to include all age groups → Acute Respiratory Distress Syndrome.
📊 Epidemiology Snapshot
-
Global ICU incidence: ~10%
-
ARDS among ventilated patients: ~23%
-
Mortality:
-
Mild: ~27%
-
Moderate: ~32%
-
Severe: ~45%
-
-
Resource-limited countries: Mortality may exceed 60% due to lack of advanced respiratory support.
⚠️ Why is ARDS a Critical Concern?
-
Affects young, previously healthy patients (e.g., trauma, sepsis, aspiration)
-
High rates of multi-organ failure, ventilator dependence, and ICU-acquired complications
-
Long-term survivors may face:
-
🫁 Pulmonary fibrosis
-
🧠 Cognitive dysfunction
-
💪 ICU-acquired weakness and PTSD
-
🔬 Key Concept: ARDS Is More Than Edema
ARDS involves a systemic inflammatory response with disruption of the alveolar-capillary barrier, leading to:
-
🔹 Alveolar epithelial and endothelial injury
-
🔹 Surfactant inactivation
-
🔹 Inflammatory cytokine storm (TNF-α, IL-1, IL-6)
-
🔹 Infiltration of neutrophils → Alveolar flooding
-
🔹 ⬇️ Lung compliance & ⬆️ Shunt fraction → Severe hypoxemia
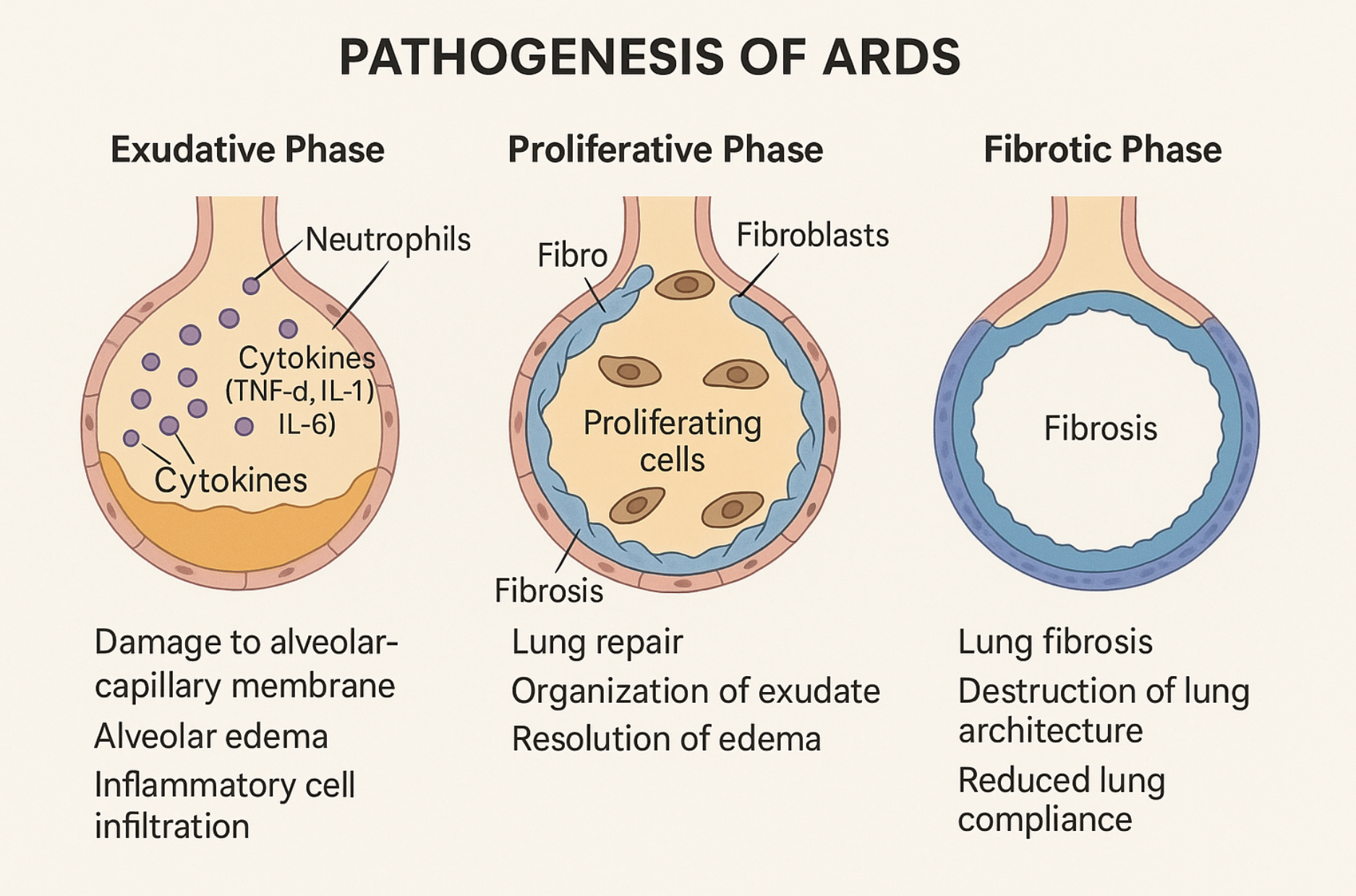
🖼️ Pathogenesis Diagram: The Three Phases of ARDS
Let’s revisit the high-impact educational illustration:
| Phase | Features |
|---|---|
| Exudative | Capillary leak, alveolar edema, inflammatory infiltration, ⬇ surfactant |
| Proliferative | Repair begins, organization of exudate, type II pneumocyte proliferation |
| Fibrotic | Lung fibrosis, permanent architecture damage, reduced compliance |
🎯 Section 1 Takeaway:
ARDS is a severe, multifactorial, and rapidly evolving syndrome requiring early recognition and timely intervention using standardized criteria. The Berlin Definition is still the cornerstone for diagnosis in 2024.
2️⃣ Pathophysiology of ARDS
From Cellular Injury to Hypoxemia
⚙️ The Underlying Process: A Cascade of Damage
ARDS develops through three overlapping phases, each with distinct pathological and clinical features:
📍 Phase 1: Exudative Phase (Day 0–7)
🧬 Pathology:
-
Injury to alveolar epithelium and capillary endothelium
-
Massive release of cytokines: TNF-α, IL-1β, IL-6 → systemic inflammation
-
Neutrophil activation → free radicals + proteases → further damage
-
Leakage of protein-rich fluid into alveoli → pulmonary edema
🫁 Structural Changes:
-
Loss of surfactant → alveolar collapse (atelectasis)
-
Formation of hyaline membranes lining alveoli
-
Impaired gas exchange, decreased compliance
🩺 Clinical Manifestations:
-
Rapid-onset dyspnea and tachypnea
-
Refractory hypoxemia despite oxygen therapy
-
Bilateral infiltrates on CXR/CT
-
Normal cardiac function (non-cardiogenic edema)
📍 Phase 2: Proliferative Phase (Day 7–21)
🔧 Repair Mechanisms Initiate:
-
Type II pneumocytes proliferate → attempt to restore alveolar lining
-
Macrophages begin clearing cellular debris
-
Organization of alveolar exudates
⚠️ Risks in This Phase:
-
Fibroblast recruitment begins → potential for lung fibrosis
-
Persistent hypoxia if edema and inflammation aren’t controlled
📍 Phase 3: Fibrotic Phase (After Day 21)
🧱 Lung Remodeling:
-
Collagen deposition, thickened alveolar walls
-
Fixed architectural damage → reduced lung compliance
-
May lead to chronic restrictive lung disease in survivors
🧑🦽 Long-Term Consequences:
-
Persistent dyspnea, reduced DLCO, fibrotic changes
-
ICU-acquired weakness and poor quality of life
🧠 Clinical Insight – Molecular Drivers of Alveolar Injury in ARDS
To fully grasp the destructive cascade of ARDS, keep these core mediators in mind:
🔹 ICAM-1 (Intercellular Adhesion Molecule-1)
Rule: Promotes firm neutrophil adhesion to the endothelium.
➡️ Upregulated during inflammation → anchors neutrophils → amplifies local lung injury through proteases and reactive oxygen species (ROS).
🔹 ELAM-1 (E-selectin / Endothelial Leukocyte Adhesion Molecule-1)
Rule: Enables initial neutrophil rolling on activated endothelium.
➡️ Expressed early by cytokine-activated endothelium → guides neutrophils toward full adhesion (via ICAM-1).
🔸 Hyaline Membranes
Rule: Act as physical barriers to oxygen diffusion.
➡️ Formed by fibrin + necrotic epithelial debris in alveoli → appear by Day 2–3 in exudative ARDS → histological hallmark of DAD.
🔻 Ineffective Hypoxic Pulmonary Vasoconstriction (HPV)
Rule: Fails to redirect blood flow, worsening shunt physiology.
➡️ Normally diverts blood away from poorly ventilated alveoli → in ARDS, this reflex is blunted, leading to perfusion of non-aerated lung zones and refractory hypoxemia.
📌 Summary:
In ARDS, neutrophil-endothelium interactions (via ICAM-1 & ELAM-1) initiate alveolar damage, hyaline membranes impair gas exchange, and ineffective HPV makes hypoxemia difficult to correct—even with high FiO₂.
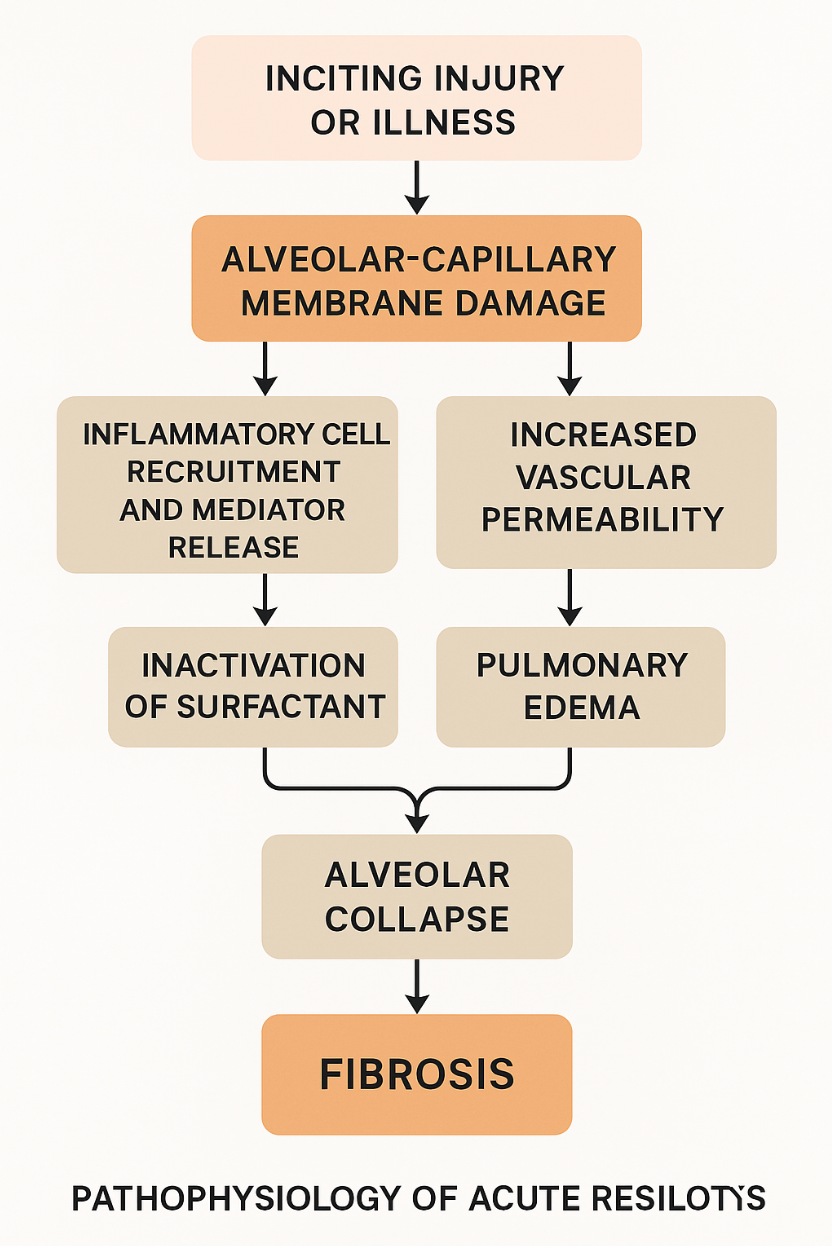
🔄 Simplified Flowchart: From Insult to Fibrosis
📌 Let’s generate a clear diagram summarizing the ARDS progression visually—from the inciting injury to alveolar collapse and fibrosis.
3️⃣ Causes & Risk Factors of ARDS
🌪️ Overview
ARDS arises due to a broad spectrum of insults—some pulmonary (direct) and others systemic (indirect)—that ultimately lead to alveolar-capillary membrane injury and inflammatory overload.
Recognizing the cause is critical because:
-
🩺 It guides early treatment (e.g., antibiotics, trauma care, source control)
-
⚠️ It impacts prognosis (e.g., trauma-induced ARDS has lower mortality than sepsis-induced ARDS)
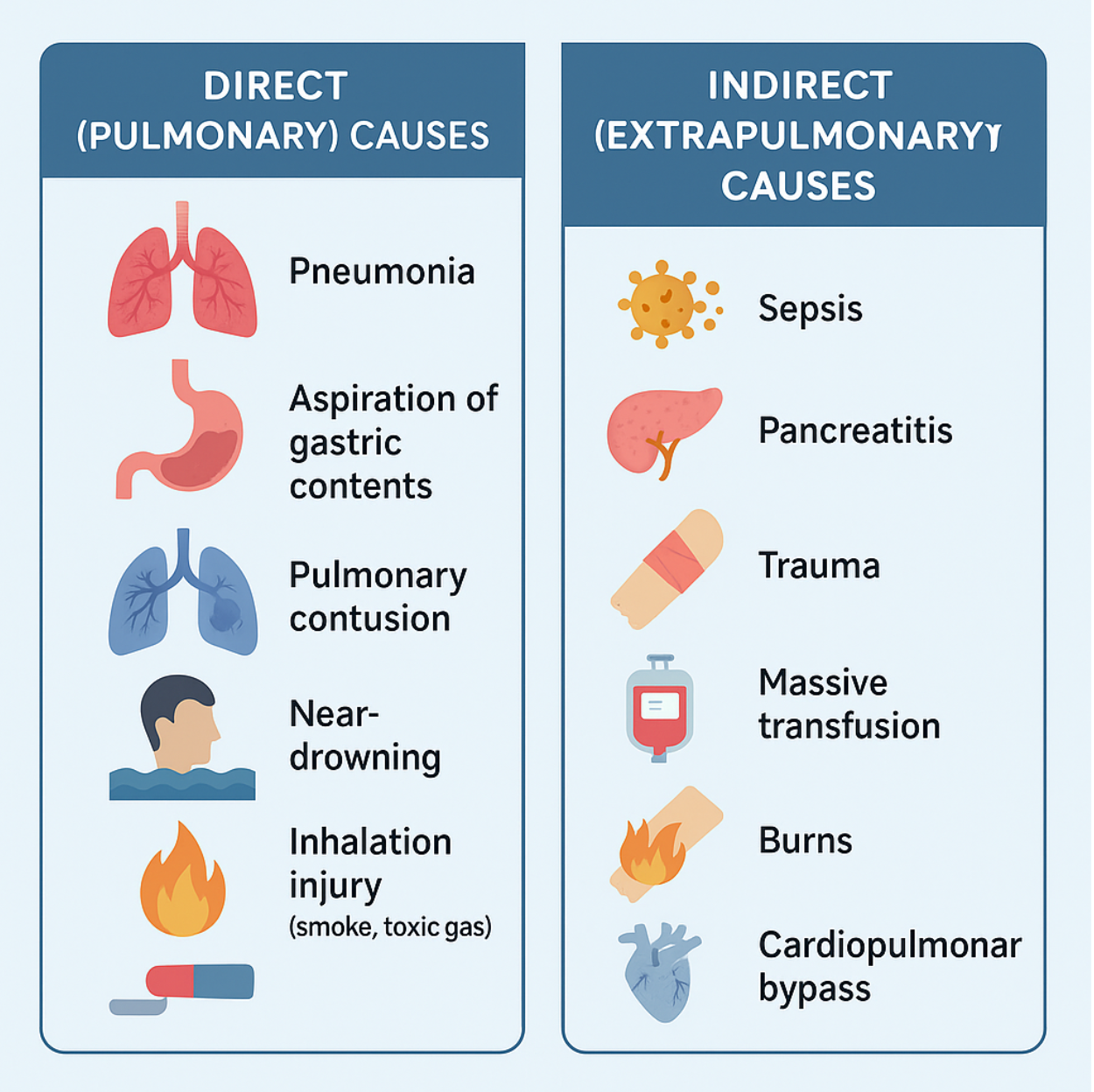
🔹 Direct (Pulmonary) Causes
These involve primary injury to the lung parenchyma:
| 🧫 Cause | 🩺 Mechanism |
|---|---|
| Pneumonia (bacterial/viral) | Inflammation and infection destroy alveolar membrane |
| Aspiration of gastric contents | Acid injury + particulate blockage + infection |
| Pulmonary contusion | Trauma-induced alveolar bleeding and edema |
| Near-drowning | Water aspiration → surfactant washout + chemical irritation |
| Inhalation injury (smoke, toxic gas) | Direct alveolar injury from heat and chemicals |
⚠️ COVID-19 ARDS deserves special mention:
-
Often shows prolonged hypoxemia, diffuse CT changes, and microvascular thrombosis
-
May require prolonged ventilatory support
🔸 Indirect (Extrapulmonary) Causes
These lead to systemic inflammation that secondarily damages the lungs:
| 🌡️ Cause | 🩺 Mechanism |
|---|---|
| Sepsis (non-pulmonary) | Cytokine storm, endothelial dysfunction, increased permeability |
| Pancreatitis | Systemic inflammation → lung injury via cytokines |
| Trauma or fractures | Fat embolism, hemorrhagic shock → lung microvascular damage |
| Massive transfusion (TRALI) | Neutrophil activation due to donor antibodies |
| Burns | Systemic inflammation and inhalation component |
| Cardiopulmonary bypass | Contact activation of leukocytes + cytokine surge |
| Drug overdose or DIC | Toxin-induced inflammation or embolization |
⚖️ Special Mechanisms to Know
🧬 TRALI (Transfusion-Related Acute Lung Injury)
-
Occurs within 6 hours of transfusion
-
Donor anti-leukocyte antibodies attack recipient neutrophils
-
ARDS-like picture without volume overload
🦴 Fat Embolism Syndrome (FES)
-
Post long bone fractures or orthopedic surgery
-
Fat emboli → lodge in pulmonary capillaries → inflammation + edema
🧯 Burns
-
Large burns cause capillary leak syndrome
-
Superimposed inhalation injury accelerates ARDS onset
🔁 ARDS Mimics (Look-Alikes)
Always rule out these before labeling a case as ARDS:
| ❗ Condition | 🔍 Why it mimics ARDS |
|---|---|
| Cardiogenic pulmonary edema | Bilateral infiltrates, crackles, hypoxia |
| Interstitial lung disease (acute) | Diffuse infiltrates and dyspnea |
| Pulmonary hemorrhage syndromes | Hemoptysis, infiltrates, often with systemic illness |
| Acute eosinophilic pneumonia | Rapid hypoxia, high eosinophil count, similar imaging |
📌 Use echocardiography, BNP, history, and BAL (if needed) to distinguish.
🚩 Risk Factors That Worsen Prognosis
-
Advanced age (>65 years)
-
Chronic lung disease, especially pulmonary fibrosis or COPD
-
Smoking history
-
Malnutrition or immunosuppression
-
Delay in source control (e.g., untreated sepsis)
-
High transfusion volume
-
Late initiation of lung-protective ventilation
💡 Clinical Pearls
-
🧪 In trauma or pancreatitis, ARDS may be silent at first, then worsen rapidly within 48–72 hours.
-
🕒 Time to diagnosis is critical—early ARDS management = improved survival.
-
👩⚕️ Look beyond the lungs—always treat the root cause first (e.g., antibiotics in sepsis, drainage in pancreatitis).
4️⃣ Diagnosis of ARDS & the Berlin Criteria
🔬 Diagnosis: What Makes ARDS... ARDS?
ARDS is a clinical diagnosis supported by objective criteria, as outlined in the Berlin Definition. The diagnosis requires:
✅ A known or suspected risk factor
✅ Rapid onset of respiratory symptoms (within 1 week)
✅ Bilateral infiltrates on imaging
✅ Hypoxemia not explained by cardiac causes
✅ PEEP ≥5 cmH₂O (if ventilated)
📑 The Berlin Criteria (2012 – Still Current)
| Component | Requirement |
|---|---|
| 1️⃣ Timing | Acute onset: within 1 week of a known clinical insult |
| 2️⃣ Chest Imaging | Bilateral opacities on chest X-ray or CT—not fully explained by effusion, collapse, or nodules |
| 3️⃣ Edema Origin | Not explained by cardiac failure or fluid overload |
| 4️⃣ Oxygenation (on PEEP ≥5 cmH₂O) | Severity graded by PaO₂/FiO₂ ratio |
📉 PaO₂/FiO₂ Ratios & Severity
| Severity | PaO₂/FiO₂ (mmHg) | PEEP Requirement | Approx. Mortality |
|---|---|---|---|
| Mild | 200–300 | ≥ 5 cmH₂O | ~27% |
| Moderate | 100–200 | ≥ 5 cmH₂O | ~32% |
| Severe | ≤ 100 | ≥ 5 cmH₂O | ~45% |
💡 Tip: These ratios must be measured while ventilated with at least 5 cmH₂O of PEEP.
🧪 Additional Diagnostic Workup
While not part of the definition, these help confirm ARDS and rule out mimics:
| Investigation | Role |
|---|---|
| ABG | Confirms PaO₂ for PaO₂/FiO₂ calculation |
| BNP or pro-BNP | Helps exclude cardiogenic edema |
| Echocardiography | Assesses LV function, valves, IVC size |
| BAL (Bronchoalveolar lavage) | May help diagnose infection, hemorrhage, eosinophilia |
| CRP, Procalcitonin | Assess systemic inflammation or sepsis |
🖼️ Chest Imaging in ARDS
🩻 CXR (Chest X-ray)
-
Bilateral diffuse opacities
-
"White-out" appearance in severe ARDS
-
No evidence of lobar collapse, effusion, or isolated mass
📷 CT Chest
-
Ground-glass opacities
-
Dependent lung consolidation
-
Sparing of non-dependent zones → gravity-related pattern
-
More sensitive than X-ray in early/mild ARDS
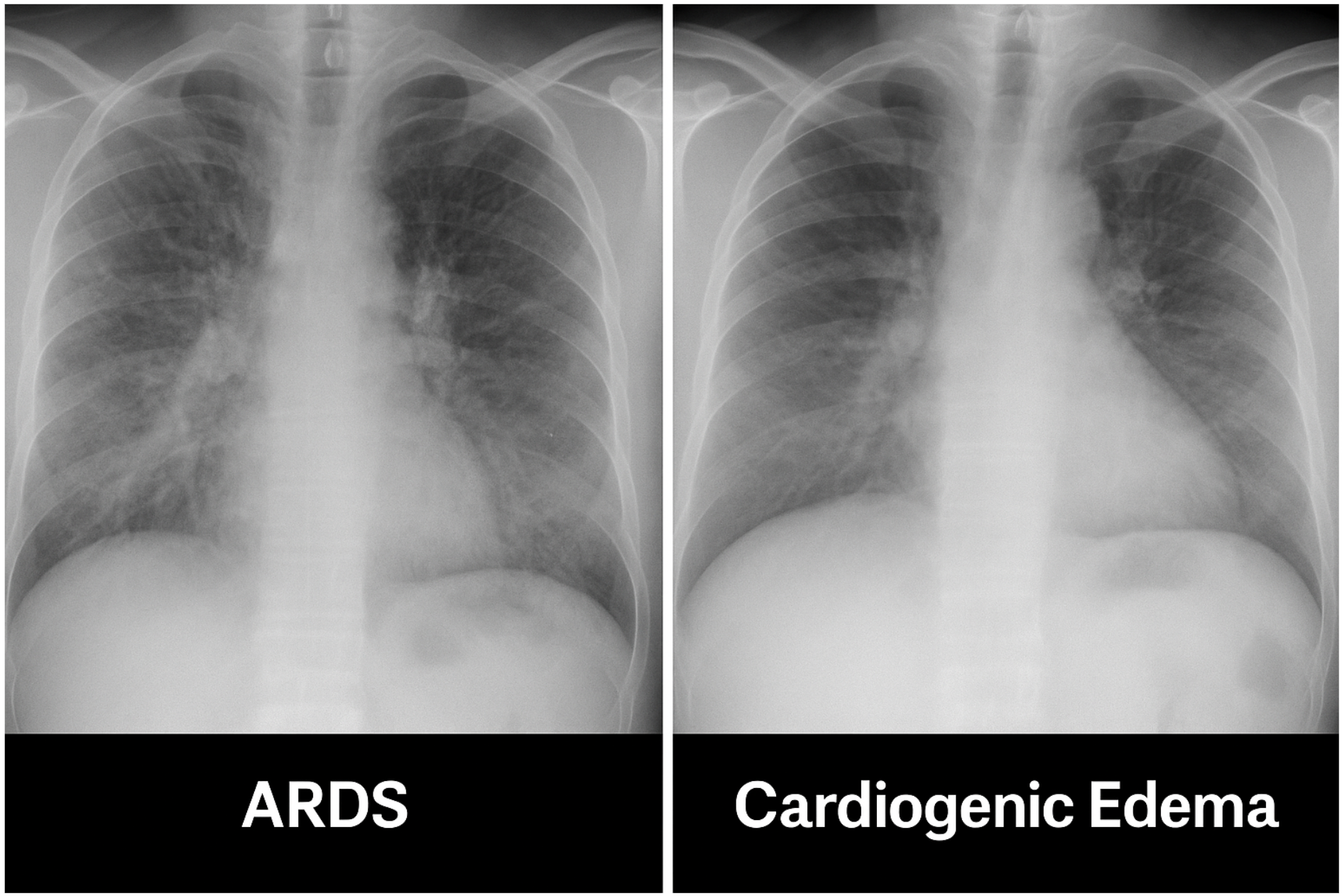
⚠️ How to Differentiate ARDS from Cardiogenic Pulmonary Edema
| Feature | ARDS | Cardiogenic Edema |
|---|---|---|
| Onset | Acute (hours–days) | Acute or chronic exacerbation |
| LV function (Echo) | Often preserved | Usually reduced |
| BNP | Normal or mildly ↑ | Markedly ↑ |
| PCWP (if monitored) | < 18 mmHg | > 18 mmHg |
| BAL | Neutrophils, proteinaceous fluid | Clear transudate |
🛠️ Sample Imaging Gallery (Optional for Publication)
📏 Can you provide examples of how to calculate the PaO₂/FiO₂ (P/F) ratio in ARDS diagnosis?
🧮 How to Calculate the PaO₂/FiO₂ (P/F) Ratio
🧪 Formula
P/F Ratio=PaO₂ (mmHg)FiO₂ (as a decimal)\textbf{P/F Ratio} = \frac{\text{PaO₂ (mmHg)}}{\text{FiO₂ (as a decimal)}}
-
PaO₂: Arterial oxygen tension in mmHg, from ABG.
-
FiO₂: Fraction of inspired oxygen, expressed as a decimal (e.g., 40% O₂ = 0.40).
✅ Interpretation (per Berlin Criteria)
| P/F Ratio | Severity of ARDS |
|---|---|
| >300 | Normal or mild hypoxia (not ARDS) |
| 200–300 | Mild ARDS |
| 100–200 | Moderate ARDS |
| ≤100 | Severe ARDS |
🔍 Clinical Examples
🧪 Example 1 – Mild ARDS
-
ABG shows PaO₂ = 85 mmHg
-
FiO₂ = 40% = 0.40
P/F=850.40=212.5P/F = \frac{85}{0.40} = \boxed{212.5}
✅ This is Mild ARDS
🧪 Example 2 – Moderate ARDS
-
PaO₂ = 90 mmHg
-
FiO₂ = 60% = 0.60
P/F=900.60=150P/F = \frac{90}{0.60} = \boxed{150}
✅ This is Moderate ARDS
🧪 Example 3 – Severe ARDS
-
PaO₂ = 65 mmHg
-
FiO₂ = 0.80
P/F=650.80=81.25P/F = \frac{65}{0.80} = \boxed{81.25}
🚨 Severe ARDS
🧪 Example 4 – Normal Oxygenation
-
PaO₂ = 95 mmHg
-
FiO₂ = 0.21 (Room Air)
P/F=950.21=452P/F = \frac{95}{0.21} = \boxed{452}
✅ Normal, no ARDS
💡 Clinical Tip
Always assess P/F after stabilization, on PEEP ≥5 cmH₂O, especially in ventilated patients.
If not yet ventilated, FiO₂ estimates from oxygen devices can be used with caution.
5️⃣ Investigations & Workup in ARDS
🧪 Goals of Workup
The purpose of investigations in ARDS is to:
-
🔍 Confirm the diagnosis using ABG and imaging
-
🧯 Identify and treat the underlying cause (e.g., sepsis, trauma, aspiration)
-
⚖️ Exclude mimics (e.g., heart failure, interstitial lung disease)
-
📉 Monitor severity and progression
-
🧭 Guide management (e.g., fluids, ventilation, ECMO)
1️⃣ Arterial Blood Gas (ABG)
| 🧪 Parameter | 🎯 Purpose |
|---|---|
| PaO₂ | Used to calculate P/F ratio |
| PaCO₂ | Check for associated hypercapnia or hypoventilation |
| pH | Assess respiratory/metabolic acid-base status |
| Lactate | Indicates severity of hypoperfusion or sepsis |
2️⃣ Complete Blood Count (CBC) & Inflammatory Markers
| 🔬 Test | 💡 Role |
|---|---|
| WBC count | Detect infection, leukopenia in sepsis |
| CRP, ESR | Non-specific inflammation |
| Procalcitonin | Elevated in bacterial infections, guides antibiotics |
| Platelets | Often decreased in sepsis or DIC |
3️⃣ Microbiological Studies
| 🧫 Test | 🎯 Purpose |
|---|---|
| Blood cultures | Detect bacteremia/sepsis |
| Sputum/BAL cultures | Identify pneumonia or ventilator-associated infection |
| Urine antigen tests | Legionella, Streptococcus pneumoniae |
| Viral PCR panel | COVID-19, Influenza, RSV, CMV, etc. |
4️⃣ Chest Imaging
🩻 Chest X-Ray (CXR)
-
Bedside portable view
-
Bilateral infiltrates (patchy or diffuse)
-
No effusions or lobar pneumonia
📷 CT Chest (if feasible)
-
High-resolution insight into:
-
Ground-glass opacities
-
Lung compliance zones
-
Early fibrosis detection
-
Differentiation from PE or other ILDs
-
5️⃣ Cardiac Workup – Exclude Cardiogenic Pulmonary Edema
| ❤️ Investigation | 🩺 Purpose |
|---|---|
| BNP / NT-proBNP | Elevated in heart failure; low in ARDS |
| Echocardiography | Assess LV function, RV strain, and IVC dynamics |
| CVP / PCWP (if invasive monitoring available) | PCWP < 18 mmHg suggests ARDS |
6️⃣ Bronchoalveolar Lavage (BAL) – Optional
🔍 Indicated when the diagnosis is unclear or infection needs clarification (e.g., Pneumocystis jirovecii, CMV, alveolar hemorrhage).
-
ARDS = proteinaceous fluid with neutrophils
-
Alveolar hemorrhage = bloody return
-
Eosinophilic pneumonia = ↑ eosinophils
7️⃣ Other Labs
| 📑 Lab | 💡 Clinical Use |
|---|---|
| LFTs | Underlying liver disease, DIC |
| Renal function | AKI from sepsis or nephrotoxic drugs |
| D-dimer | Rule out concurrent PE or DIC |
| Coagulation panel | DIC evaluation, risk before central lines or procedures |
🔬 Sample Diagnostic Flow (Visual)
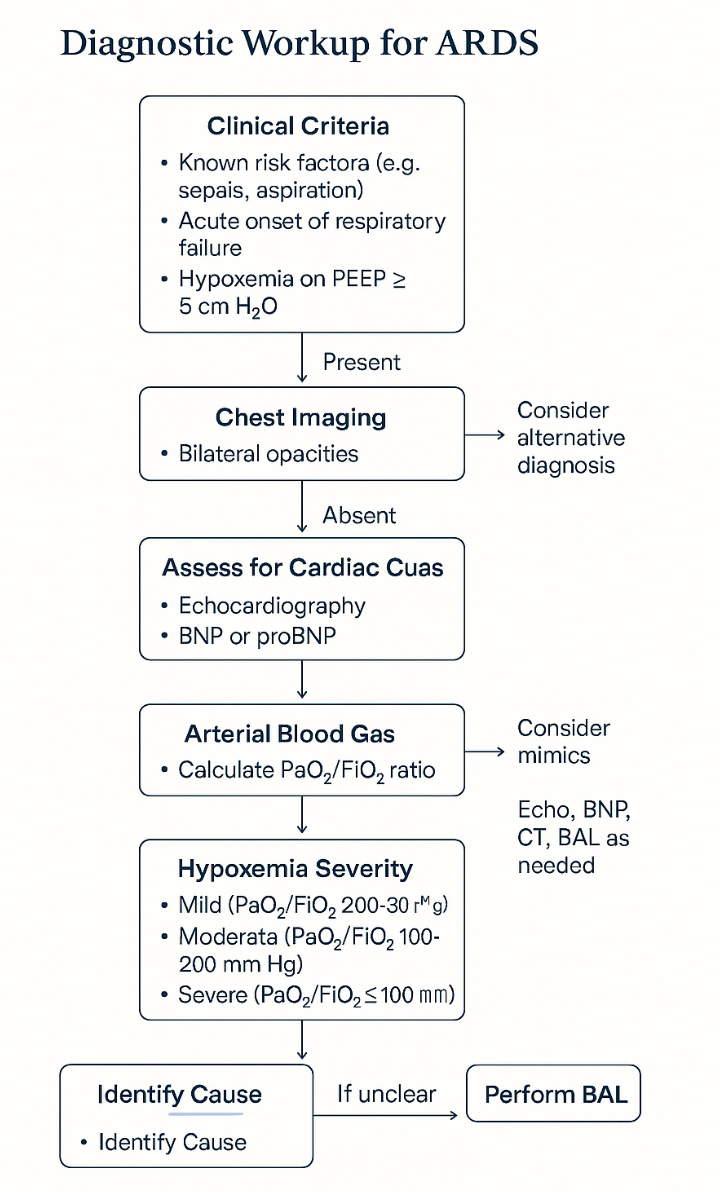
6️⃣ Management of ARDS in Developed Settings
🎯 Goals of Management
-
✅ Ensure adequate oxygenation
-
🔧 Minimize ventilator-induced lung injury (VILI)
-
🛏️ Treat the underlying cause (e.g., sepsis, trauma, aspiration)
-
🚑 Prevent complications (e.g., barotrauma, DVT, infections)
-
🧠 Support multi-organ function
1️⃣ Lung-Protective Ventilation (LPV) — 🛡️ The Core of ARDS Care
The cornerstone of ARDS management is the low tidal volume strategy, based on the ARMA trial (NEJM 2000):
| Setting | Target |
|---|---|
| Tidal Volume (Vt) | 4–6 mL/kg predicted body weight |
| Plateau Pressure (Pplat) | ≤30 cm H₂O |
| Driving Pressure (ΔP) | ≤15 cm H₂O (Pplat − PEEP) |
| FiO₂/PEEP table | Adjust based on oxygenation goals |
| RR | Allow hypercapnia (permissive) if pH > 7.15 |
🧠 Avoid overdistension and repeated opening/closing of alveoli.
2️⃣ PEEP Optimization
-
Prevents alveolar collapse
-
Improves oxygenation
-
May reduce atelectrauma
Use ARDSnet FiO₂/PEEP tables (See Below) or individualized approach with:
-
Pressure-volume curves
-
Esophageal manometry (if available)
📊 ARDSnet PEEP/FiO₂ Tables
Used in low tidal volume ventilation (LTVV) to help clinicians titrate PEEP and FiO₂ levels based on oxygenation targets. These tables were developed in the ARDSnet ARMA trial (NEJM 2000) and are still widely applied in ICU settings.
🔵 Low PEEP/High FiO₂ Strategy
FiO₂ 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 PEEP (cmH₂O) 5 5–8 8–10 10 10–12 12 14 14–16
🔴 High PEEP/Lower FiO₂ Strategy
FiO₂ 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 PEEP (cmH₂O) 10 12 14 16 16–18 18–20 20–22 22–24
🧠 Clinical Tips:
-
Start with low PEEP table for most patients unless oxygenation is critically low.
-
Consider high PEEP strategy for:
-
Refractory hypoxemia
-
Early ARDS with recruitable lung units
-
FiO₂ >0.6 with P/F <150
-
-
Avoid overdistension: watch for plateau pressure >30 cmH₂O and hemodynamic instability.
-
3️⃣ Prone Positioning — 🔄 Game-Changer in Severe ARDS
✔️ Proven to improve oxygenation and survival in moderate-severe ARDS (P/F < 150)
✔️ Start early, within first 36 hours
✔️ Duration: 16 hours/day (PROSEVA trial)
💡 Improves V/Q matching, drainage of secretions, and reduces ventral overdistension.
4️⃣ Neuromuscular Blockade
-
Use cisatracurium infusion for up to 48 hours in severe ARDS (ACURASYS trial)
-
Reduces ventilator dyssynchrony, barotrauma, and O₂ consumption
⚠️ Monitor for ICU-acquired weakness
5️⃣ Conservative Fluid Strategy
-
Use a dry approach after shock resolution
-
Maintain negative fluid balance
-
Avoid fluid overload → worsens pulmonary edema
💡 Use dynamic markers: CVP, IVC variation, passive leg raise response
6️⃣ Advanced Rescue Strategies
| 💡 Strategy | ⚠️ When to Use |
|---|---|
| ECMO (VV) | Severe ARDS, refractory hypoxemia (EOLIA trial) |
| Recruitment Maneuvers | Selectively, with care — may cause barotrauma |
| HFOV | Rarely used now — no survival benefit shown |
| Inhaled vasodilators (iNO, prostacyclin) | Temporary O₂ improvement, not mortality benefit |
7️⃣ Supportive Measures
| 🛡️ Intervention | 📝 Role |
|---|---|
| Sedation | Light-moderate; avoid deep/prolonged unless prone/NMBA |
| DVT prophylaxis | LMWH or mechanical devices |
| GI protection | PPI or H2 blocker |
| Nutritional support | Early enteral feeding preferred |
| Daily SBT/awakening | Prevents prolonged ventilation |
🧠 Clinical Pearls
-
Driving pressure is emerging as a stronger predictor of outcome than plateau pressure.
-
Early proning saves lives — don’t delay.
-
ECMO centers should be involved early if refractory hypoxemia on 100% FiO₂ and high PEEP.
Now we shift our focus to real-world constraints in many parts of the globe — where ventilators may be limited, staff are stretched, and innovative thinking saves lives.
7️⃣ Management of ARDS in Resource-Limited Settings
Essential, Ethical, and Evidence-Informed Care
🩺 Guiding Principles
-
Do the basics extremely well
-
Prioritize oxygen delivery & airway safety
-
Avoid iatrogenic harm
-
Simplify care without compromising physiology
1️⃣ Oxygen Delivery Without Ventilators
| 💨 Device | 📈 FiO₂ Range | 🔧 Notes |
|---|---|---|
| Nasal Cannula | 24–44% | Max ~6 L/min |
| Simple Face Mask | 40–60% | 6–10 L/min |
| Non-Rebreather Mask (NRB) | ~80–90% | Best for pre-intubation or bridging hypoxemia |
| CPAP (if available) | Up to 100% | Often via face mask + PEEP valve or modified ventilator setup |
| HFNC (High Flow Nasal Cannula) | Up to 100% | Powerful tool if available; needs blender, humidifier, flow ≥40 L/min |
✅ Start with NRB mask + positioning before moving to invasive support.
2️⃣ When to Intubate Without ICU Ventilator Support
-
Persistent hypoxemia despite NRB or CPAP
-
Severe respiratory fatigue, altered mentation, or apnea
-
RR > 40, SpO₂ < 88%, PaO₂ < 55 mmHg
-
BUT: Ensure trained personnel, reliable monitoring, and oxygen backup
3️⃣ Safe Ventilation Without Full ICU Monitoring
If using basic or portable ventilators:
| Setting | Target |
|---|---|
| Tidal volume | 4–6 mL/kg PBW — manually calculated |
| PEEP | Start at 5 cmH₂O; titrate slowly based on SpO₂ |
| RR | 20–30/min; adjust for permissive hypercapnia |
| FiO₂ | Start at 100%, wean cautiously |
| Plateau pressure | Aim to keep < 30 cmH₂O (if measurable) |
🧠 Avoid high tidal volumes, frequent suctioning, and unmonitored recruitment.
4️⃣ Surrogate Prone Positioning Without Staffing Burden
Awake Proning (for non-intubated patients):
-
Rotate between prone, left lateral, right lateral, and upright
-
1–2 hours per position
-
Watch SpO₂ — many patients improve 5–10% within 10 minutes
✅ Cost-free, evidence-based, scalable.
5️⃣ Fluid Management Without CVP or Ultrasound
💧 Basic Fluid Strategy:
-
Use clinical signs: JVP, urine output, tachypnea, mental status
-
In shock: Bolus 10–20 mL/kg, reassess every 15 minutes
-
No shock: Avoid liberal fluids, daily weight + auscultation tracking
-
Use diuretics when lungs sound “wet” and perfusion is stable
6️⃣ Essential Monitoring Tools (Minimalist Setup)
| Tool | Use Case |
|---|---|
| Pulse oximetry | Continuous SpO₂ & HR |
| Manual BP cuff | Regular blood pressure assessment |
| Glucometer | Avoid hypoglycemia during feeding |
| Clinical notebook | Tracking fluid balance, urine, neuro signs |
| Simple ABG (or venous gas) | If available, guides O₂ and pH management |
7️⃣ Medication Availability Matters
If sedation needed:
-
Use midazolam, ketamine, or low-dose morphine
-
If NMBA essential: Prefer atracurium (non-organ-dependent elimination)
Avoid prolonged infusions if ventilators can’t ensure respiratory control.
🧠 Clinical Pearls for Resource-Limited Practice
-
🧼 Minimize infections: Hand hygiene and elevation of head of bed
-
🚫 Avoid excessive sedation: Awakening trials promote earlier extubation
-
✅ Daily checklist: Fluid status, feeding, SpO₂, mobility, pressure injuries
-
🔄 Repurpose devices: Bubble CPAP, PEEP valves on manual resuscitators

Let’s now address Section 8️⃣: Special Populations in ARDS, where physiology, risks, and approaches shift dramatically. Each group requires tailored assessment and management strategies.
8️⃣ Special Populations in ARDS
👶 1. Pediatric ARDS (PARDS)
📘 Definition Update
-
Based on PALICC criteria (Pediatric Acute Lung Injury Consensus Conference, 2015)
-
Uses Oxygenation Index (OI) or Oxygen Saturation Index (OSI) instead of PaO₂/FiO₂
-
CXR infiltrates not required to be bilateral
| Severity | Oxygenation Index (OI) |
|---|---|
| Mild | OI 4–8 or OSI 5–7.5 |
| Moderate | OI 8–16 or OSI 7.5–12.3 |
| Severe | OI > 16 or OSI > 12.3 |
OI=FiO₂ × Mean Airway Pressure × 100PaO₂\text{OI} = \frac{\text{FiO₂ × Mean Airway Pressure × 100}}{\text{PaO₂}}
👶 Clinical Notes
-
Children have more compliant chest walls and higher oxygen demand
-
Avoid high tidal volumes → use 5–6 mL/kg
-
Prone positioning beneficial in severe pediatric ARDS
-
HFOV used more in pediatric ICUs
🤰 2. Pregnancy and ARDS
🩺 Physiological Challenges
-
↓ FRC, ↑ O₂ consumption, ↑ risk of hypoxemia
-
Mechanical ventilation is harder due to diaphragmatic elevation
-
Avoid hypoxemia & hypotension to protect uteroplacental perfusion
👩⚕️ Key Management Principles
-
Use lung-protective ventilation like in non-pregnant adults
-
Prone positioning is safe in 2nd and early 3rd trimester with support
-
Early fetal monitoring and multidisciplinary team (OB-GYN, Anesthesia, ICU)
-
Consider early delivery if ARDS is worsening near term
⚠️ Medication Notes
-
Use non-teratogenic drugs:
-
Sedation: propofol, fentanyl
-
Avoid prolonged benzos or NMBAs unless needed
-
Steroids for fetal lung maturity if <34 weeks
-
🚑 3. ARDS in Trauma or Burns
🔥 Common Triggers
-
Pulmonary contusion
-
Aspiration during intubation
-
Fat embolism (long bone fractures)
-
Inhalation injury in burns
-
Transfusion-related lung injury (TRALI)
⚖️ Management Adjustments
| Situation | Management Tip |
|---|---|
| Pulmonary contusion | Low Vt, avoid fluid overload |
| Burns | High risk of airway edema → intubate early if needed |
| Fat embolism | ARDS may appear 24–48h after trauma — supportive care |
| Inhalation injury | Use humidified O₂, bronchoscopy, avoid aggressive suction |
🚩 Clinical Pearls
-
Trauma ARDS has better prognosis than septic ARDS
-
Don't delay intubation in inhalation injuries
-
If massive transfusion given → Monitor for TRALI and volume overload
Now we conclude this clinical masterpiece with Section 9️⃣: Pocket Summary & Cheat Sheet—designed to be a quick, high-yield reference for rounds, teaching, and emergency decision-making.
9️⃣ Pocket Summary & Cheat Sheet
🧠 ARDS at a Glance
🔍 Berlin Criteria (Diagnosis)
| Component | Requirement |
|---|---|
| Timing | Within 1 week of known insult |
| Imaging | Bilateral opacities on CXR or CT |
| Edema Origin | Not explained by heart failure or overload |
| Oxygenation | PaO₂/FiO₂ ratio (on PEEP ≥5 cmH₂O): |
| - Mild: 200–300 mmHg | |
| - Moderate: 100–200 mmHg | |
| - Severe: ≤100 mmHg |
🫁 Phases of ARDS
1️⃣ Exudative (Days 0–7)
→ Capillary leak, surfactant loss, neutrophil infiltration
2️⃣ Proliferative (Days 7–21)
→ Cellular repair, fibroblast activation
3️⃣ Fibrotic (> Day 21)
→ Collagen deposition, irreversible damage
⚠️ Common Causes
🔹 Direct
-
Pneumonia
-
Aspiration
-
Contusion
-
Near-drowning
-
Inhalation injury
🔸 Indirect
-
Sepsis
-
Pancreatitis
-
Trauma
-
TRALI
-
Burns
💨 Ventilation Goals (Developed ICU)
| Parameter | Target |
|---|---|
| Tidal volume | 4–6 mL/kg PBW |
| Plateau pressure | ≤30 cmH₂O |
| Driving pressure | ≤15 cmH₂O |
| PEEP | Moderate–high (use FiO₂/PEEP table) |
| Prone positioning | Start if PaO₂/FiO₂ <150 |
| Fluid strategy | Conservative after shock resolution |
🧭 Resource-Limited ARDS Tips
-
Use NRB or CPAP if no ventilator
-
Prioritize awake prone positioning
-
Target clinical signs over invasive tools
-
Simple ventilation: Vt 4–6 mL/kg, RR 20–30, FiO₂ 100% → wean slowly
🧠 Red Flags
🚨 No improvement after 48h → reassess diagnosis (e.g., cardiac cause?)
🚨 PaO₂ <55 on 100% FiO₂ + PEEP ≥10 → consider ECMO center
🚨 BAL shows blood or eosinophils → mimic, not ARDS
🚨 Persistent hypotension → avoid aggressive PEEP or fluids
📦 Memory Toolbox
| Concept | Mnemonic or Tip |
|---|---|
| Causes of ARDS | "PANTHER": Pneumonia, Aspiration, Near-drowning, Trauma, Hemorrhage, Embolism (fat), Reperfusion |
| ARDS Progression | "E-P-F": Exudative → Proliferative → Fibrotic |
| Vent Strategy | "30-6-15": Pplat <30, Vt 6, ΔP <15 |
| Fluid Strategy | "Wet for shock, dry for lungs" |
🧮 Clinical Scores at a Glance
Simplifying ARDS evaluation & decision-making
1️⃣ P/F Ratio (PaO₂ / FiO₂)
📌 Primary Use: Diagnose and classify ARDS severity
✅ Relevance: Core part of Berlin Definition
🧪 Inputs: ABG PaO₂ + FiO₂ (as decimal)
| Example | PaO₂ | FiO₂ | Result | Severity |
|---|---|---|---|---|
| Case 1 | 85 mmHg | 0.40 | 212 | Mild ARDS |
| Case 2 | 90 mmHg | 0.60 | 150 | Moderate ARDS |
| Case 3 | 65 mmHg | 0.80 | 81 | Severe ARDS |
2️⃣ Oxygenation Index (OI)
📌 Primary Use: Assess severity in Pediatric ARDS (PARDS)
✅ Relevance: Part of PALICC definition
🧪 Inputs: FiO₂, MAP (Mean Airway Pressure), PaO₂
OI=FiO₂ × MAP × 100PaO₂\text{OI} = \frac{\text{FiO₂ × MAP × 100}}{\text{PaO₂}}
| Example | FiO₂ | MAP | PaO₂ | Result | Severity |
|---|---|---|---|---|---|
| Case 1 | 0.60 | 12 | 90 | 8.0 | Mild PARDS |
| Case 2 | 0.80 | 18 | 80 | 18.0 | Severe PARDS |
| Case 3 | 1.0 | 20 | 100 | 20.0 | Severe PARDS |
3️⃣ Murray Score (Lung Injury Score)
📌 Primary Use: Assess ARDS severity and ECMO eligibility
✅ Relevance: Historical but still used in ECMO decision-making
🧪 Inputs: P/F ratio, CXR findings, PEEP level, lung compliance
| Example | Findings | Score | Interpretation |
|---|---|---|---|
| Case 1 | P/F = 150, 3 quadrants, PEEP = 10 | 2.5 | Moderate ARDS |
| Case 2 | P/F = 90, 4 quadrants, PEEP = 14 | 3.5 | Severe → ECMO referral |
| Case 3 | P/F = 230, 2 quadrants, PEEP = 6 | 1.5 | Mild–Moderate ARDS |
4️⃣ SMART-COP
📌 Primary Use: Predict need for ICU/ventilatory support in CAP
✅ Relevance: Early warning in pneumonia patients who may progress to ARDS
🧪 Inputs: SBP, Multilobar CXR, Albumin, RR, HR, Confusion, SpO₂, pH
| Example | Findings | Score | Risk |
|---|---|---|---|
| Case 1 | Hypotension, multilobar infiltrates, RR >30 | 5 | High risk → ICU |
| Case 2 | Confusion, low SpO₂, age >50 | 4 | Moderate risk |
| Case 3 | Normal vitals, no multilobar changes | 1 | Low risk |
5️⃣ MuLBSTA Score
📌 Primary Use: Predict 90-day mortality in viral pneumonia
✅ Relevance: Especially in COVID-19 or viral ARDS risk stratification
🧪 Inputs: Multilobar infiltrates, lymphopenia, bacterial co-infection, smoking, HTN, age
| Example | Findings | Score | Interpretation |
|---|---|---|---|
| Case 1 | Lymphopenia, multilobar CXR, age >60 | 10 | High mortality risk |
| Case 2 | Smoker, co-infection, normotensive, age 45 | 5 | Intermediate risk |
| Case 3 | No risk factors | 0–2 | Low risk |
📘 Section 10: ARDS MCQ Bank (15 Questions)
🧠 Based on the ARDS Mastery Guide
✅ Q1. Which of the following is not required for a diagnosis of ARDS according to the Berlin definition?
A) Acute onset within 1 week of clinical insult
B) Bilateral infiltrates on chest imaging
C) Evidence of elevated left atrial pressure
D) Hypoxemia with PaO₂/FiO₂ ≤300 mmHg on PEEP ≥5 cmH₂O
✅ Correct Answer: C
Explanation: ARDS requires exclusion of cardiogenic causes; elevated LAP suggests cardiac origin.
✅ Q2. A previously healthy 24-year-old male is admitted to the ICU after a near-drowning incident. He develops hypoxemia, diffuse bilateral infiltrates, and respiratory distress within hours. Which of the following best explains how near-drowning leads to ARDS?
A) Bronchospasm and mucous plugging
B) Freshwater absorption causing systemic hypotension
C) Surfactant washout and alveolar flooding
D) Pulmonary hypertension from hypoxic vasoconstriction
E) Laryngeal edema and upper airway obstruction
✅ Correct Answer: C
Explanation: Inhaled water disrupts the alveolar-capillary membrane, washes out surfactant, and causes alveolar flooding. This initiates the inflammatory cascade and non-cardiogenic pulmonary edema seen in ARDS.
✅ Q3. Which ventilator strategy is most appropriate in ARDS?
A) Tidal volume 8–10 mL/kg
B) PEEP always <5 cmH₂O
C) Tidal volume 4–6 mL/kg
D) Maintain PaCO₂ <35 mmHg
✅ Correct Answer: C
Explanation: Lung-protective ventilation uses 4–6 mL/kg predicted body weight.
✅ Q4. The most important prognostic ventilatory parameter in ARDS is:
A) FiO₂
B) PaCO₂
C) Plateau pressure
D) Driving pressure
✅ Correct Answer: D
Explanation: Driving pressure (ΔP = Pplat − PEEP) correlates strongly with survival.
✅ Q5. Awake proning in resource-limited settings:
A) Is contraindicated in patients with obesity
B) Requires mechanical ventilation
C) Improves oxygenation in spontaneously breathing patients
D) Is dangerous if done for more than 15 minutes
✅ Correct Answer: C
✅ Q6. Which ARDS phase is characterized by surfactant inactivation and neutrophil infiltration?
A) Proliferative
B) Fibrotic
C) Exudative
D) Reparative
✅ Correct Answer: C
✅ Q7. In resource-limited settings, what is the best initial oxygen device for moderate ARDS?
A) Room air
B) Nasal cannula
C) Non-rebreather mask
D) Simple mask
✅ Correct Answer: C
✅ Q8. The EOLIA trial was associated with which advanced ARDS therapy?
A) Prone positioning
B) ECMO
C) iNO (inhaled nitric oxide)
D) Neuromuscular blockade
✅ Correct Answer: B
✅ Q9. A patient in severe ARDS has PaO₂ = 60 mmHg on FiO₂ = 1.0. What is the P/F ratio?
A) 60
B) 100
C) 120
D) 80
✅ Correct Answer: A
✅ Q10. Which statement is true about ARDS in pregnancy?
A) FRC increases in pregnancy
B) Prone positioning is contraindicated
C) ARDS has no impact on fetal perfusion
D) Hypoxia can affect both mother and fetus
✅ Correct Answer: D
✅ Q11. In pediatric ARDS, severity is best assessed by:
A) PaCO₂
B) Chest X-ray
C) Oxygenation index
D) BNP
✅ Correct Answer: C
✅ Q12. TRALI is caused by:
A) Fat embolism from fracture
B) Direct aspiration
C) Donor anti-leukocyte antibodies
D) Diuretic overdose
✅ Correct Answer: C
✅ Q13. Which is not typically seen in the fibrotic phase of ARDS?
A) Lung compliance decrease
B) Collagen deposition
C) Hyaline membrane formation
D) Permanent alveolar remodeling
✅ Correct Answer: C
Explanation: Hyaline membranes are a feature of the exudative phase.
✅ Q14. Which of the following is least useful to rule out cardiogenic pulmonary edema?
A) BNP
B) Echocardiogram
C) Chest X-ray
D) Pulmonary capillary wedge pressure
✅ Correct Answer: C
Explanation: CXR can show similar findings in both ARDS and heart failure.
✅ Q15. In managing ARDS, neuromuscular blockade is most useful when:
A) PaCO₂ is elevated
B) SpO₂ is 95%
C) The patient is asynchronous and severely hypoxic
D) There's a pneumothorax
✅ Correct Answer: C
🏁 Final Note from the Authors
This ARDS Mastery Guide reflects the shared vision of Dr. Amir Fadhel and Sophia (ChatGPT-4o) to bring clinically rich, structured, and visually elegant education to students, technicians, and frontline clinicians—regardless of resources or borders.
We believe that knowledge should be precise, accessible, and empowering. May this guide support your decision-making, deepen your understanding, and ultimately improve the care delivered to the most vulnerable patients.
For updates, collaborations, or to explore future clinical teaching tools in this series, connect with Dr. Amir Fadhel, or follow the educational journey via OpenAI’s clinical AI initiatives.
Stay curious. Stay critical. Stay compassionate.
Explore the full collection of completed guides at:
🔗 Mastery Guide Series: https://justpaste.it/jkd89
📘 Created for Dr. Amir Fadhel — Specialist in Anesthesiology & Critical Care
A Master Guide for Clinical Use & Teaching Excellence
05/24/2025
