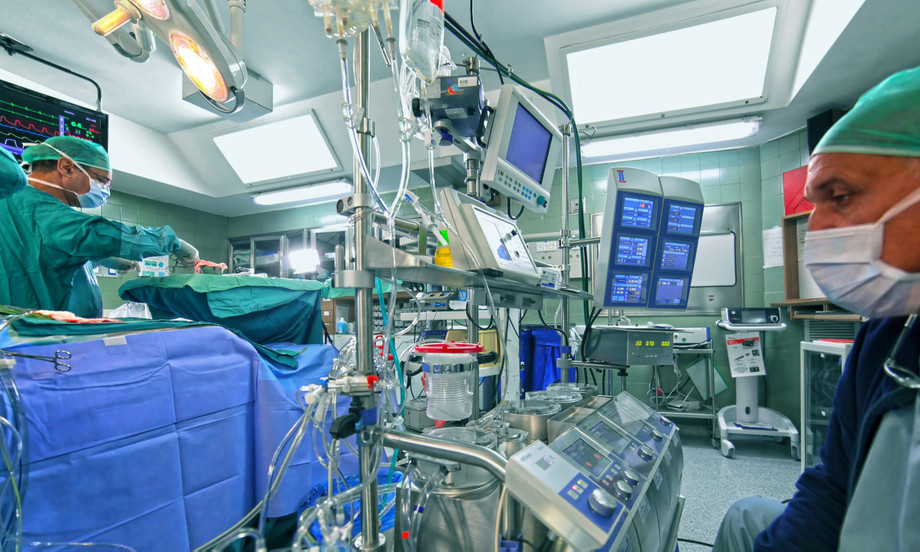
Cardiopulmonary bypass equipment comprises heart–lung machines, membrane oxygenators, centrifugal and roller pumps, heat exchangers, and advanced monitoring systems designed to take over cardiac and pulmonary function during open-heart surgery. These devices offer precise regulation of blood flow, optimal gas exchange, temperature control, and real-time monitoring, significantly reducing perioperative risks and enhancing patient safety. Growing demand for minimally invasive cardiac procedures, coupled with an aging population prone to cardiovascular ailments, has driven R&D investments and improved design ergonomics. Manufacturers leverage automated perfusion technologies to streamline clinical workflow, improve market share, and address stringent regulatory requirements.
Cardiopulmonary Bypass Equipment Market is estimated to be valued at USD 210.9 Mn in 2025 and is expected to reach USD 256.0 Mn in 2032, exhibiting a compound annual growth rate (CAGR) of 2.8% from 2025 to 2032.
Key Takeaways
Key players operating in the Cardiopulmonary Bypass Equipment Market are Medtronic, LivaNova (formerly Sorin Group), Terumo Cardiovascular Systems Corporation, Maquet Holding BV & Co. KG, Braun Melsungen AG, Medica s.p.a., Eurosets, Fresenius Medical Care AG & Co. KGaA, Lepu Medical Technology, Elite Life Care, OriGen Biomedical, Tianjin Medical, Soma Technology, Inc., Braile Biomédica, CardioGenesis Corporation, Century Medical, Inc., Nonin Medical, Inc., Xijian Medical. These market companies leverage extensive R&D pipelines, strategic alliances, and robust distribution networks to maintain leadership in product innovation, regulatory compliance, and service support.
Growing Demand
Cardiopulmonary Bypass Equipment Market Demand is rising due to an increase in cardiac surgeries like bypass grafts and valve replacements. Aging populations and higher cardiovascular disease rates sustain this growth. The trend toward minimally invasive and robotic-assisted procedures also drives need for compact, automated systems. Supportive reimbursement policies, improved healthcare infrastructure, and greater clinician awareness of perfusion practices further accelerate adoption, creating strong opportunities for both established firms and new entrants.
Global Expansion
North America leads the market with advanced healthcare systems and high surgical volumes. Europe follows, supported by strong government backing for cardiac care. Asia Pacific is growing rapidly thanks to improved healthcare access, rising elective surgeries, and collaborations with global players. Latin America and the Middle East & Africa offer future potential as healthcare investments grow and cardiac care expands, reshaping the global competitive landscape.
Market Key Trends
A major trend is the integration of digital perfusion and real-time monitoring into bypass systems. Predictive analytics and IoT-enabled consoles are enhancing surgical outcomes by optimizing hemodynamic monitoring. AI is aiding perfusionists in making data-driven decisions during operations. As patient safety gains focus, manufacturers are investing in device interoperability and cybersecurity, driving innovation and shaping long-term market strategies.
Porter’s Analysis
Threat of new entrants: High levels of capital investment and technical expertise are required to design, develop and validate heart–lung machine components in compliance with rigorous international regulatory frameworks and quality management systems, reflecting the complex market dynamics that discourage newcomers. Moreover, anticipated investment for establishing reliable sales networks, clinical trial partnerships and aftermarket service support dissuades smaller firms from contesting established incumbents, limiting fresh competition.
Bargaining power of buyers: Large hospitals and integrated health systems wield significant negotiating leverage due to consolidated purchasing volumes and long-term supply agreements for cardiopulmonary bypass disposables and equipment, enabling them to drive price concessions, safeguard service-level commitments and heighten market challenges for manufacturers. However, switching costs remain elevated given the critical importance of validated compatibility with existing perfusion protocols and maintenance infrastructure, which tempers buyer influence amid specialized clinical requirements.
Bargaining power of suppliers: Providers of specialized medical-grade polymers, microfilters and biomedical sensors maintain moderate to high influence over cost structures and supply continuity for cardiopulmonary bypass equipment manufacturers, especially when offering proprietary materials with limited alternative sources. Long lead times for certification and long-term contracts for consumable kits further restrict manufacturers’ flexibility to negotiate raw material pricing, creating potential supply-side constraints in production planning and affecting market insights around sourcing strategies.
Threat of new substitutes: Emerging extracorporeal life support technologies, such as portable ECMO and minimally invasive cardiac assist devices, present potential alternatives to traditional heart–lung machine circuits in certain perioperative and critical care scenarios, though their application remains confined to specialized case profiles and acute settings. Additionally, progression in artificial organ development and regenerative medicine could gradually reduce reliance on conventional perfusion systems for select patient populations, opening niche market opportunities over the long term.
Competitive rivalry: Intense competition among regional and global medical device companies drives continuous innovation in pump efficiency, oxygenator design and digital monitoring features to capture greater market share and sustain business growth.
Geographical Regions: Market Value Concentration
The cardiopulmonary bypass equipment market is dominated by North America and Western Europe, which account for the majority of global revenue. North America's strong healthcare infrastructure, high cardiac surgery volumes, and favorable reimbursement policies drive demand. Continuous investment in cardiac centers and upgrades to perfusion labs further strengthen market growth. Europe follows closely, with Germany, France, the UK, and Italy leading the way. The region benefits from mature regulations and the growing use of minimally invasive techniques. Market research and clinical workshops also help shape future product development.
Asia Pacific has a smaller share but sees significant adoption in Japan and Australia. Healthcare improvements in China and South Korea are boosting access, though reimbursement and infrastructure challenges limit full market potential. Southeast Asia and Latin America contribute modestly, impacted by public health priorities and limited capital. The Middle East & Africa, with its uneven healthcare spending, has the smallest share. Manufacturers monitor these regions to adapt to market trends and address regional barriers.
Geographical Regions: Fastest Growing Region
Asia Pacific is the fastest-growing region for cardiopulmonary bypass equipment, driven by increasing cardiovascular disease rates and expanding healthcare infrastructure. China is leading with major hospital construction, improved funding for cardiac centers, and growing private hospitals investing in advanced perfusion technology. India’s market growth is fueled by a rising middle class, broader insurance coverage, and more cardiac specialty hospitals. Japan and South Korea continue to grow with hospital upgrades and digital perfusion monitoring adoption.
Southeast Asian countries like Singapore, Malaysia, and Thailand are also seeing rapid growth, supported by medical tourism and government incentives. Regional partnerships help navigate regulatory challenges and address workforce shortages. The focus on remote monitoring and tele-perfusion modules further drives demand. Manufacturers tailor strategies to capitalize on these opportunities, mitigate supply chain risks, and fuel growth in this dynamic region.
Get This Report in Japanese Language -人工心肺装置市場
Get This Report in Korean Language -심폐우회술 장비 시장
Read More Articles Related to this Industry –
Nanofiber Applications in Medical Devices: Revolutionizing Healthcare
Camera Modules in Medical Devices: Revolutionizing Diagnostics and Treatment
About Author:
Priya Pandey is a dynamic and passionate editor with over three years of expertise in content editing and proofreading. Holding a bachelor's degree in biotechnology, Priya has a knack for making the content engaging. Her diverse portfolio includes editing documents across different industries, including food and beverages, information and technology, healthcare, chemical and materials, etc. Priya's meticulous attention to detail and commitment to excellence make her an invaluable asset in the world of content creation and refinement.
(LinkedIn- https://www.linkedin.com/in/priya-pandey-8417a8173/)