📘 Antimicrobial Therapy in the ICU — Mastery Guide
Prepared for Dr. Amir Fadhel — Specialist in Anesthesiology and Critical Care
Developed in collaboration with Sophia (ChatGPT-4o)
Referencing the acclaimed Mastery Series: ABG • Shock • Ventilation • ARDS • Sepsis • Electrolytes • DKA • PCAS
🔗 Mastery Series Collection
🧭 About This Guide
This guide is designed for clinicians navigating the critical decision-making of antibiotic use in intensive care — from initial empiric coverage to culture-based refinement, from device-related infections to resistance challenges, and from resource-rich ICUs to field hospitals with limited drug availability.
You will learn to:
🔹 Understand antimicrobial pharmacology and organ impact
🔹 Choose the right antibiotic for the right site of infection
🔹 Master timing, de-escalation, and duration strategies
🔹 Handle ICU-acquired infections like VAP, CLABSI, and CAUTI
🔹 Avoid common toxicity and resistance errors
🔹 Implement rational therapy in low-resource settings
This Mastery Guide follows our structured approach:
- Step-by-step clarity
- Clinical integration
- Visual explanations
- Red flags, pearls, and simplified checklists
Each section is formatted to be bedside-ready, teaching-friendly, and resource-conscious.
📖 Table of Contents
1️⃣ About This Guide
2️⃣ Introduction — Why Antibiotics Matter More in the ICU
3️⃣ Principles of Antimicrobial Stewardship in Critical Care
4️⃣ Empiric vs Definitive Therapy: Timing, Cultures, & Coverage
5️⃣ Pharmacokinetics & Pharmacodynamics in the Critically Ill
🔹 Dosing in renal/liver failure
🔹 Time- vs concentration-dependent killing
🔹 Tissue penetration in sepsis
🔹 Drug level monitoring (e.g., vancomycin, aminoglycosides)
6️⃣ Site-Specific Infections & ICU Antibiotic Choices
6.1 Ventilator-Associated Pneumonia (VAP) & HAP
6.2 Bloodstream Infections & CLABSI
6.3 Urinary Tract Infections & CAUTI
6.4 CNS Infections in ICU (meningitis, ventriculitis, shunt infections)
6.5 Intra-abdominal & Post-op Infections
6.6 Skin, Soft Tissue, and Necrotizing Infections
7️⃣ ICU-Acquired Infections: Red Flags & Response
🔸 Prevention bundles overview
🔸 Common pathogens & resistance profile
🔸 Duration & device management
🔸 Role of source control
8️⃣ De-escalation, Duration, and Stopping Therapy
🔸 When to narrow, how to stop
🔸 Biomarkers (e.g., Procalcitonin, CRP)
🔸 Short vs extended course strategies
9️⃣ Multidrug-Resistant Organisms (MDROs) in the ICU
🔹 ESBL, CRE, MRSA, VRE, MDR Acinetobacter
🔹 Combination therapies
🔹 Salvage regimens
🔹 Infection control coordination
🔟 Antimicrobial Use in Limited-Resource Settings
🔸 Rational antibiotic selection when labs are unavailable
🔸 Cost-effective coverage
🔸 Generic alternatives to reserve drugs
🔸 Clinical diagnosis without imaging
1️⃣1️⃣ Special Populations in the ICU
🔹 Neutropenic fever
🔹 Burn patients
🔹 Transplant/immunosuppressed
🔹 Pregnancy & lactation
1️⃣2️⃣ Practical Infographic: ICU Antibiotic Start–Stop Flowchart
1️⃣3️⃣ Pocket Summary: Empiric Antibiotic Table by Site
1️⃣4️⃣ Advanced Clinical MCQs — ICU Antibiotics in Action
1️⃣5️⃣ Final Words
1️⃣ Introduction — Why Antibiotics Matter More in the ICU
“In the ICU, antibiotics don’t just treat — they rescue, reshape outcomes, and prevent collapse.”
🌍 The ICU Is a Battlefield of Bugs and Resistance
Critically ill patients are not ordinary patients:
- They have weakened immune systems
- They’re exposed to invasive devices (ETT, CVCs, Foley)
- They often lack clear infection sources at presentation
- They experience organ dysfunction that alters drug handling
- They are at high risk of ICU-acquired infections and drug-resistant organisms
Antibiotics in the ICU are not a routine prescription — they are a high-stakes decision.
🧠 The Problem We Face
| ICU Issue | Why Antibiotics Are Complicated |
|---|---|
| Hemodynamic instability | Alters organ perfusion → impacts drug absorption and elimination |
| Renal/hepatic dysfunction | Affects drug clearance and toxicity risk |
| Altered volume of distribution | Sepsis causes leaky capillaries → underdosing if not adjusted |
| Need for empiric therapy | Cultures often not available before treatment |
| Multidrug-resistant pathogens | Colonization is common; over-treatment is dangerous |
| Limited diagnostics in some ICUs | Empiric decisions must be made with clinical skill |
📊 The Scope of Antibiotic Use in the ICU
- 60–70% of ICU patients receive antibiotics during admission
- 30–50% of ICU antibiotic use is unnecessary, inappropriate, or misdosed
- Antibiotic misuse is associated with:
🔺 Increased mortality
🔺 Clostridioides difficile infection
🔺 Rising resistance
🔺 Longer ICU stay & cost
🧪 Example: The Cost of a Mistake
A 72-year-old woman with pneumonia on day 5 of mechanical ventilation is started empirically on vancomycin + piperacillin-tazobactam. Cultures later grow MSSA, and she continues vancomycin for 10 days. She develops AKI, requires dialysis, and dies of urosepsis 2 weeks later.
The right drug (nafcillin/oxacillin) would have reduced toxicity and improved outcomes.
🧠 Clinical Wisdom:
In the ICU, we don't just ask "What antibiotic?" — we ask "Why? When? How long? What organ function? What bug risk?"
🔍 What This Guide Will Empower You To Do
- Think site-specifically: Lung? Urine? Abdomen? Brain?
- Start empirically — but de-escalate responsibly
- Choose based on PK/PD, not just brand names
- Recognize red flags for resistance
- Adapt therapy for limited-resource settings
- Avoid the two extremes: overkill vs undercoverage
2️⃣ Principles of Antimicrobial Stewardship in Critical Care
“Antibiotics save lives — but only when used wisely, not widely.”
🔎 What Is Antimicrobial Stewardship?
A systematic, evidence-based approach to ensure that the right antibiotic is used:
👉 At the right time
👉 At the right dose
👉 For the right duration
👉 In the right patient
In critical care, stewardship is not about restriction, it’s about precision — saving the patient while preserving antibiotics for future ones.
⚠️ Why Is Stewardship Essential in the ICU?
| ICU Reality | Stewardship Role |
|---|---|
| Empiric broad-spectrum use is common | Stewardship ensures timely de-escalation |
| Sepsis demands early treatment | But duration and spectrum must be reassessed |
| Resistance spreads fast in ICUs | Stewardship reduces overuse & colonization |
| Diagnostics may be delayed or absent | Clinical judgment + protocols needed to guide therapy |
| Drug toxicity is under-monitored | Stewardship includes PK/PD monitoring & dose adjustment |
🧠 Core Stewardship Strategies in the ICU
🔹 1. Start smart, then focus
- Early empiric therapy for sepsis is life-saving
- Always document indication + plan to reassess at 48–72 hrs
🔹 2. Tailor based on cultures & sensitivity
- Narrow spectrum when culture results are back
- Stop agents if no infection or colonization only
🔹 3. Optimize dosing & route
- Adjust for renal/liver function
- Choose IV vs oral wisely
🔹 4. Shorten duration
- Prolonged therapy increases resistance risk
- Use 5–7 day courses for many ICU infections if stable and improving
🔹 5. Know your local antibiogram
- Empiric choices should reflect local ICU resistance patterns
🔹 6. Use biomarkers when possible
- Procalcitonin and CRP can help with de-escalation and stopping
📌 Stewardship in Action
| Scenario | Stewardship Action |
|---|---|
| VAP with negative cultures at 72 hrs | Stop antibiotics if no clinical deterioration |
| Blood cultures grow MSSA | Switch from vancomycin to nafcillin/oxacillin |
| Stable UTI patient improving at day 5 | Stop antibiotics — do not extend to 10–14 days |
| Severe AKI with vancomycin | Trough monitoring, or switch to safer alternative |
| Febrile ICU patient post-op, no source | Reassess for non-infectious fever, do not extend empiric abx blindly |
🔬 ICU Stewardship Is Not a Luxury — It’s a Life-Saving System
🧠 It improves outcomes
🧠 Reduces mortality
🧠 Protects antibiotics for tomorrow
🧠 Helps prevent ICU-acquired infections
🧭 Frameworks to Guide ICU Antibiotic Stewardship
“Principles are good. But structure wins at the bedside.”
In the complex ICU setting, the following clinical mnemonics help translate stewardship into daily, teachable, and auditable practice — especially for residents, pharmacists, and staff working in systems with limited oversight.
✅ The 5 R’s of Antimicrobial Stewardship
| R | Clinical Application |
|---|---|
| Right Drug | Target the most likely pathogen for the infection site (e.g., Pseudomonas in VAP, MRSA in CLABSI). |
| Right Dose | Adjust for renal/hepatic function, severity of illness, and site penetration. |
| Right Route | Start IV in unstable or septic patients; switch to oral only when stable with good absorption. |
| Right Time | Administer within 1 hour of recognizing sepsis; avoid unnecessary delay for cultures. |
| Right Duration | Avoid fixed 10–14 days; use evidence-based durations (e.g., 5–7 days for pneumonia with improvement). |
✅ The 4 D’s of Antimicrobial Therapy
| D | Why It Matters |
|---|---|
| Right Drug | Chosen based on likely pathogens and site pharmacology |
| Right Dose | ICU patients have altered volume of distribution → underdosing is dangerous |
| Right Duration | Overuse breeds resistance; shorter durations often equally effective |
| Right De-escalation | After 48–72 hrs, stop or narrow spectrum once cultures and response are available |
🧠 Teaching Tip for Ward Rounds:
"Ask your team on day 3 of antibiotics — what's the R or D we're applying today?"
If no one knows, the plan is passive… not stewardship.
📎 With these two frameworks, you now have a bedside-ready language to guide daily decisions — structured, teachable, auditable.
3️⃣ Empiric vs Definitive Therapy — Timing, Cultures & Coverage
“Start wide. Get smart. Then stop.”
🔍 Why This Section Matters
In the ICU, delays in antibiotics for sepsis can kill, but failure to de-escalate or stop can also harm.
🔹 Empiric therapy saves lives in early sepsis
🔹 Definitive therapy protects against resistance and toxicity
This section helps clinicians master when to start, what to cover, and how to tailor — with practical emphasis on cultures, coverage logic, and timely reassessment.
⏱️ When to Start Antibiotics in the ICU
| Scenario | Target Timing |
|---|---|
| Septic shock or organ failure | Within 1 hour of recognition (bundle-compliant) |
| Suspected sepsis but stable | Within 3 hours |
| Non-life-threatening infection | Within 6 hours based on site & severity |
| Colonization without infection | ❌ Do not treat |
| Viral or non-infectious cause likely | Withhold antibiotics unless deterioration occurs |
🧠 Red Flag: Even with sepsis, do not delay therapy for culture collection beyond 45–60 min. Prioritize both speed and precision.
🧪 Cultures Before Antibiotics — But Don’t Wait Too Long
Always aim to collect at least:
- 🔹 Two blood cultures from separate sites
- 🔹 Urine, sputum, CSF, or relevant drainage
- 🔹 Line tip, wound, or pleural/ascitic fluid if available
📌 If unable to get cultures in <60 minutes, start empiric therapy anyway.
📦 Principles of Empiric Therapy in the ICU
Empiric antibiotics must be:
🔹 Broad-spectrum enough to cover likely pathogens
🔹 Based on infection site, local resistance, and patient risk factors
🔹 Paired wisely (e.g., avoid duplicate Gram-negative or MRSA overuse)
🔹 Adjusted to organ function and penetration needs
🧠 Example: Empiric Coverage Logic
Suspected VAP on day 7 in a ventilated patient
- Likely pathogens: Pseudomonas, MRSA, ESBLs
- Empiric combo: Piperacillin-tazobactam + Vancomycin
- If colonized with MDR Acinetobacter → consider Polymyxins
- If renal dysfunction → adjust dose or use inhaled aminoglycosides
🔬 Transitioning to Definitive Therapy
At 48–72 hours, reassess:
✅ Culture & sensitivity results
✅ Patient’s clinical course
✅ Lab and radiologic response
✅ Procalcitonin or inflammatory marker trends (if available)
✅ Key Actions at 48–72 Hours:
| Finding | Action |
|---|---|
| Positive cultures | Narrow spectrum to specific sensitive agent |
| No growth + improving | Consider stopping or narrowing |
| No growth + worsening | Broaden, investigate source, repeat cultures |
| Colonizers only | Stop antibiotics unless invasive signs |
| High procalcitonin + worsening | Escalate or reassess source control |
🧠 De-escalation is as important as starting. It saves kidneys, money, and lives.
💉 Duration Is Not Fixed — It’s Dynamic
| Infection Type | Typical ICU Duration (if improving) |
|---|---|
| Uncomplicated VAP/HAP | 7 days |
| CLABSI with no complications | 7–10 days |
| CAUTI | 5–7 days |
| Intra-abdominal source controlled | 4–7 days |
| Meningitis or deep-seated focus | 10–14+ days depending on organism |
| Fungal infections | Variable — follow clinical & lab markers |
🧾 Essential Abbreviations & Their Clinical Significance
📌 VAP — Ventilator-Associated Pneumonia
An infection of the lower respiratory tract occurring ≥48 hours after intubation.
🔹 Pathogens: Pseudomonas, MRSA, Acinetobacter
🔹 Risk: Often multi-drug resistant in ICU
📌 HAP — Hospital-Acquired Pneumonia
Pneumonia developing ≥48 hours after admission in non-ventilated patients.
🔹 Common organisms: Klebsiella, E. coli, Enterobacter
📌 CLABSI — Central Line-Associated Bloodstream Infection
Defined as bloodstream infection in a patient with a central line in place >2 days, with no other source.
🔹 Often caused by: Staphylococcus aureus, CoNS, Candida
📌 CAUTI — Catheter-Associated Urinary Tract Infection
UTI in a catheterized patient after 2 days of catheter insertion.
🔹 Often involves E. coli, Proteus, Pseudomonas
📌 ESBL — Extended-Spectrum Beta-Lactamase Producing Bacteria
Enzymes produced by E. coli, Klebsiella, making them resistant to penicillins and cephalosporins.
🔹 Often require: carbapenems or beta-lactamase inhibitor combinations
📌 CRE — Carbapenem-Resistant Enterobacteriaceae
Enterobacteriaceae resistant to all beta-lactams including carbapenems.
🔹 Therapy may include: Polymyxins, tigecycline, or combination regimens
📌 MRSA — Methicillin-Resistant Staphylococcus aureus
A Gram-positive cocci resistant to beta-lactams.
🔹 Treated with: Vancomycin, Linezolid, or Daptomycin (except in pneumonia)
📌 MSSA — Methicillin-Sensitive Staphylococcus aureus
Sensitive to oxacillin, nafcillin, and cefazolin — preferred over vancomycin when confirmed.
📌 Procalcitonin (PCT)
A biomarker elevated in bacterial infections.
🔹 Useful for de-escalating antibiotics in respiratory infections and sepsis
🔹 Decline by ≥80% = potential signal to stop therapy
📌 PK/PD — Pharmacokinetics & Pharmacodynamics
- PK = How the body handles the drug (absorption, distribution, metabolism, excretion)
- PD = How the drug affects the organism (time- or concentration-dependent killing)
🧠 In ICU, both PK and PD are profoundly altered — so doses must be customized, not standardized.
📎 Takeaway:
“Start wide. Document cultures. Reassess early. Stop early. Trust response more than routine.”
4️⃣ Pharmacokinetics & Pharmacodynamics in the Critically Ill
“In the ICU, antibiotics don’t move the same, don’t kill the same — and don’t forgive mistakes.”
💡 Why This Matters
In critical illness, your patient’s body:
🔹 Leaks fluids
🔹 Alters drug clearance
🔹 Has low albumin
🔹 Is sometimes on dialysis or ECMO
That means standard doses won’t work — they can be ineffective or toxic.
📊 What Is Pharmacokinetics (PK)?
“What the body does to the drug.”
| Component | ICU Impact |
|---|---|
| Absorption | May be reduced in shock or gut edema |
| Distribution | ↑ Volume of distribution (Vd) in sepsis → lower drug levels |
| Metabolism | Slowed in hepatic dysfunction |
| Excretion | Altered by AKI, CRRT, ECMO → dose adjustment crucial |
🔬 What Is Pharmacodynamics (PD)?
“What the drug does to the organism.”
Different antibiotics kill bacteria in different ways:
| Antibiotic Class | Killing Type | Strategy |
|---|---|---|
| Beta-lactams | ⏳ Time-dependent | Keep levels above MIC as long as possible (T>MIC) |
| Aminoglycosides | 💥 Concentration-dependent | High peak is critical (Cmax/MIC) |
| Fluoroquinolones | ⚖️ AUC-dependent | Need total exposure over time (AUC/MIC) |
| Vancomycin | 🎯 AUC-guided | Target AUC > 400 (mg·h/L) for best efficacy |
🧠 MIC = Minimum Inhibitory Concentration
It’s the lowest drug level needed to stop bacterial growth.
⚠️ ICU PK/PD Challenges in Real Life
| Clinical Situation | Effect on PK/PD |
|---|---|
| Sepsis or burns | ↑ Vd → may need higher loading doses |
| Hypoalbuminemia | ↓ protein binding → ↑ free drug → ↑ effect/toxicity |
| Renal failure or dialysis | ↓ clearance → risk of accumulation |
| CRRT / ECMO | Drug is removed or bound in circuit → dosing changes needed |
| Obesity or fluid overload | May require weight-based adjustment for aminoglycosides, vancomycin |
🧪 Practical Clinical Examples
1️⃣ Piperacillin–Tazobactam in Sepsis
– Time-dependent
– Use extended infusion (4 hours)
→ Keeps drug above MIC longer = better kill
2️⃣ Gentamicin in VAP
– Concentration-dependent
– Give high-dose once daily
→ Maximizes peak (Cmax) and allows washout to reduce nephrotoxicity
3️⃣ Vancomycin in AKI
– Renally cleared
– Use loading dose (25–30 mg/kg), then adjust based on AUC or trough
– Avoid overdosing in reduced clearance
📌 ICU Dosing Pearls
| Antibiotic | ICU Consideration |
|---|---|
| Meropenem | Give as 3-hr extended infusion |
| Vancomycin | Target AUC > 400, or trough 15–20 mg/L |
| Colistin | Nephrotoxic — dose carefully in AKI |
| Linezolid | No renal adjustment needed, but monitor for myelosuppression |
| Aminoglycosides | Use ideal body weight, monitor peaks & troughs |
🧠 Clinical Wisdom:
“In critical illness, drug levels lie — trust physiology, not the package insert.”
📋 ICU Antibiotic Dosing Quick Table
Standard doses, loading strategies, and key ICU adjustments
| Antibiotic | Standard ICU Dose | ICU Tips & Adjustments |
|---|---|---|
| Piperacillin–Tazobactam | 4.5 g IV q6h or 4.5 g over 4h q8h (extended) | Use extended infusion to increase T>MIC |
| Meropenem | 1 g IV q8h or 2 g q8h (for severe) | Use 3-hr infusion; renal dose adjust |
| Cefepime | 2 g IV q8h | Consider neurotoxicity in renal failure |
| Ceftriaxone | 1–2 g IV q24h | No renal adjustment needed |
| Vancomycin | 25–30 mg/kg IV load, then 15–20 mg/kg q8–12h | Monitor AUC or trough; dose by actual body weight |
| Linezolid | 600 mg IV/PO q12h | No renal adjustment; watch for myelosuppression |
| Gentamicin | 5–7 mg/kg IV once daily (extended interval) | Monitor peak (8–10 mg/L) and trough (<1 mg/L) |
| Amikacin | 15–20 mg/kg IV once daily | Adjust for obesity & renal function |
| Colistin (CMS) | 9 MU IV loading, then 4.5 MU q12h | Watch for nephro- and neurotoxicity |
| Tigecycline | 100 mg load, then 50 mg q12h | Poor bloodstream penetration |
| Fluconazole | 800 mg IV load, then 400 mg q24h | Good CNS penetration |
| Metronidazole | 500 mg IV/PO q8h | Hepatic clearance — caution in liver disease |
| Ciprofloxacin | 400 mg IV q8–12h | Use AUC target; adjust in renal dysfunction |
| Levofloxacin | 750 mg IV q24h | QTc monitoring if on other QT-prolonging drugs |
🧠 Symbols to Remember:
- 💉 Extended infusion = better for beta-lactams (T>MIC)
- ⚠️ Monitor levels = aminoglycosides & vancomycin
- 🚫 Avoid in renal failure = colistin, cefepime (toxicity risk)
- 🩸 Avoid in thrombocytopenia = linezolid, prolonged beta-lactams
💎 ICU Antibiotic Dosing Pearls — Pocket Favorites
| Antibiotic | Dosing Pearl |
|---|---|
| Ceftriaxone | 1–2 g IV q24h (↑ to 2 g q12h in meningitis); no renal adjustment needed 💧 |
| Meropenem | 1–2 g q8h via 3-hour infusion 🕒 — ideal for ESBLs & CNS infections 🧠 |
| Piperacillin–Tazobactam | Use extended 4-hour infusion → better time above MIC ⏳ |
| Vancomycin | Load 25–30 mg/kg; target AUC > 400 or trough 15–20 mg/L 🎯 |
| Gentamicin | 5–7 mg/kg once daily; monitor Cmax (8–10) and trough (<1) 🎧 |
🧠 These aren’t just drugs — they’re tactical weapons. Dose them with intent, love, and a touch of wisdom.
🧾 Abbreviations & ICU Antibiotic Concepts — Master Glossary
This is your quick-access reference for understanding the language of dosing, monitoring, and precision care in ICU antimicrobial therapy.
🩸 Cmax (Maximum Plasma Concentration)
- The peak drug level in the blood after administration
- High Cmax = stronger bacterial kill for concentration-dependent antibiotics
- Relevant for: Gentamicin, Amikacin, Fluoroquinolones
🎯 “Aim high to strike hard.”
🩸 Trough (Minimum Drug Level)
- The lowest drug concentration before the next dose
- Keeping trough low avoids toxicity
- Crucial in: Gentamicin (nephrotoxicity), Vancomycin (traditional method)
🛡 “Clear it before next strike — protect the kidneys.”
🩸 AUC (Area Under the Curve)
- The total exposure to drug over time
- Best marker of drug effectiveness + safety in many antibiotics
- Ideal for: Vancomycin, Linezolid, Fluoroquinolones
💡 “The full 24-hour story — not just peak or trough.”
🧪 MIC (Minimum Inhibitory Concentration)
- The lowest concentration of antibiotic that inhibits visible bacterial growth
- All dosing targets (Cmax, AUC) are compared against MIC
- Helps determine if an antibiotic can effectively treat an infection
⚖️ “Know your enemy’s weakness — MIC tells you that.”
⚙️ T>MIC (Time Above MIC)
- Key concept for time-dependent antibiotics (e.g., beta-lactams)
- Drug must stay above MIC for most of the dosing interval
- Strategy: Use extended or continuous infusions
⏳ “Keep the drug present — not just strong.”
💊 Cmax/MIC, AUC/MIC
- Ratios used to evaluate drug killing potential
- Higher ratio = better chance of bacterial eradication
- For gentamicin: Cmax/MIC ≥ 8–10
- For vancomycin: AUC/MIC ≥ 400
5️⃣ Site-Specific Infections & ICU Antibiotic Choices
“The source decides the sword.”
🧭 Why This Section Matters
In the ICU, knowing where the infection is matters as much as knowing who the pathogen is.
🔹 Some sites need blood-brain barrier penetration
🔹 Some harbor anaerobes
🔹 Some are full of biofilms (catheters, prosthetics)
🔹 And some — like lungs or intra-abdominal spaces — require combination therapy and source control
This section maps each site, its likely culprits, and the best antibiotic approach — always with ICU considerations.
📌 ICU Site-Specific Infections — Common Pathogens & Empiric Antibiotics
| Infection Site | Likely Pathogens | First-Line Empiric Therapy | ICU Notes |
|---|---|---|---|
| Lungs (VAP/HAP) | Pseudomonas, MRSA, Klebsiella, Acinetobacter | Pip–Tazo or Meropenem + Vancomycin or Linezolid | Use double anti-pseudomonal in high-risk MDR; reassess at 72h |
| CNS (Meningitis, abscess) | Strep pneumo, Neisseria, Listeria (age), GNB, HSV | Ceftriaxone 2g q12h + Vancomycin ± Ampicillin (if Listeria risk) ± Acyclovir (if viral) | Need agents with CNS penetration; avoid vancomycin monotherapy |
| Urinary Tract (CAUTI) | E. coli, Klebsiella, Enterococcus, Pseudomonas | Cefepime or Meropenem ± Vancomycin | Adjust for ESBL/CRE; de-escalate if culture allows |
| Bloodstream (CLABSI) | Staph aureus (MRSA/MSSA), Candida, GNB | Vancomycin + Cefepime or Meropenem ± Echinocandin (if fungal risk) | Remove central line early; repeat cultures; de-escalate after sensitivities |
| Intra-abdominal (IAI) | Enterobacteriaceae, Bacteroides, Enterococcus | Pip–Tazo or Meropenem ± Metronidazole | Source control is critical; don’t just escalate antibiotics |
| Skin/Soft Tissue | Strep pyogenes, Staph aureus (MRSA/MSSA), anaerobes | Vancomycin + Pip–Tazo (or Meropenem for necrotizing fasciitis) | Consider surgical debridement in necrotizing infections |
| Catheter-related Sepsis | CoNS, Staph aureus, Candida | Vancomycin ± Echinocandin or Cefepime (if septic) | Remove catheter; consider fungal if patient on TPN or immunosuppressed |
| Biliary Tract / Liver | Enterobacteriaceae, Enterococcus, anaerobes | Ceftriaxone + Metronidazole or Pip–Tazo | Surgical source control may be needed (e.g., drainage of cholangitis or abscess) |
| Endocarditis | Strep viridans, Staph aureus, Enterococcus, HACEK | Vancomycin + Ceftriaxone (empiric) | Tailor after culture; monitor embolic and valve complications |
🧠 Clinical Tips:
- Always ask: Is the source controlled? If not, antibiotics alone will fail.
- Use local antibiograms where available
- Step down to narrow-spectrum within 72h if patient improves and cultures guide you
- Don’t miss fungal coverage in TPN, prolonged ICU, or broad-spectrum prior use
🫁 Lung Infections in the ICU — VAP & HAP
“The lungs are the frontlines — infection strikes here first.”
🧠 What Are VAP and HAP?
| Term | Definition |
|---|---|
| HAP | Pneumonia ≥48 hrs after admission, not present at admission |
| VAP | Pneumonia ≥48 hrs after intubation and mechanical ventilation |
🦠 Common Pathogens in VAP/HAP
| Organism | Clinical Relevance |
|---|---|
| Pseudomonas aeruginosa | Common, MDR risk especially >5 days in ICU |
| Staphylococcus aureus (MRSA) | Risk in diabetics, CKD, prolonged hospital stay |
| Klebsiella, Enterobacter, E. coli | ESBL risk in prior antibiotics or immunosuppressed |
| Acinetobacter spp. | Highly resistant — endemic in some ICU settings |
💊 Empiric Antibiotic Strategy
| Risk Level | Empiric Treatment |
|---|---|
| Low MDR risk | Pip–Tazo 4.5 g IV q6h or Cefepime 2 g q8h |
| High MDR risk | Meropenem 1–2 g IV q8h + Vancomycin or Linezolid |
| Suspected MRSA | Add Vancomycin (AUC-based) or Linezolid 600 mg IV q12h |
| Suspected ESBL or CRE | Consider Meropenem, or Polymyxins if CRE confirmed |
🧠 Always consider local antibiogram and recent culture data
🧪 Collect ET aspirate or BAL if possible before antibiotics
⏱ Timing Is Critical
- Start antibiotics within 1 hour if patient is septic or in shock
- Collect cultures before, but don’t delay antibiotics >1 hour
📅 Duration of Therapy
| Clinical Course | Duration | Notes |
|---|---|---|
| Clinical improvement | 7 days | If source controlled, no MDR, no complications |
| MDR or slow recovery | 10–14 days | Tailor to organism & patient status |
🧠 Red Flags in VAP/HAP
- No improvement in 72h? Think: Wrong bug, wrong drug, or wrong source
- Always reassess: CXR, oxygenation, cultures, inflammatory markers

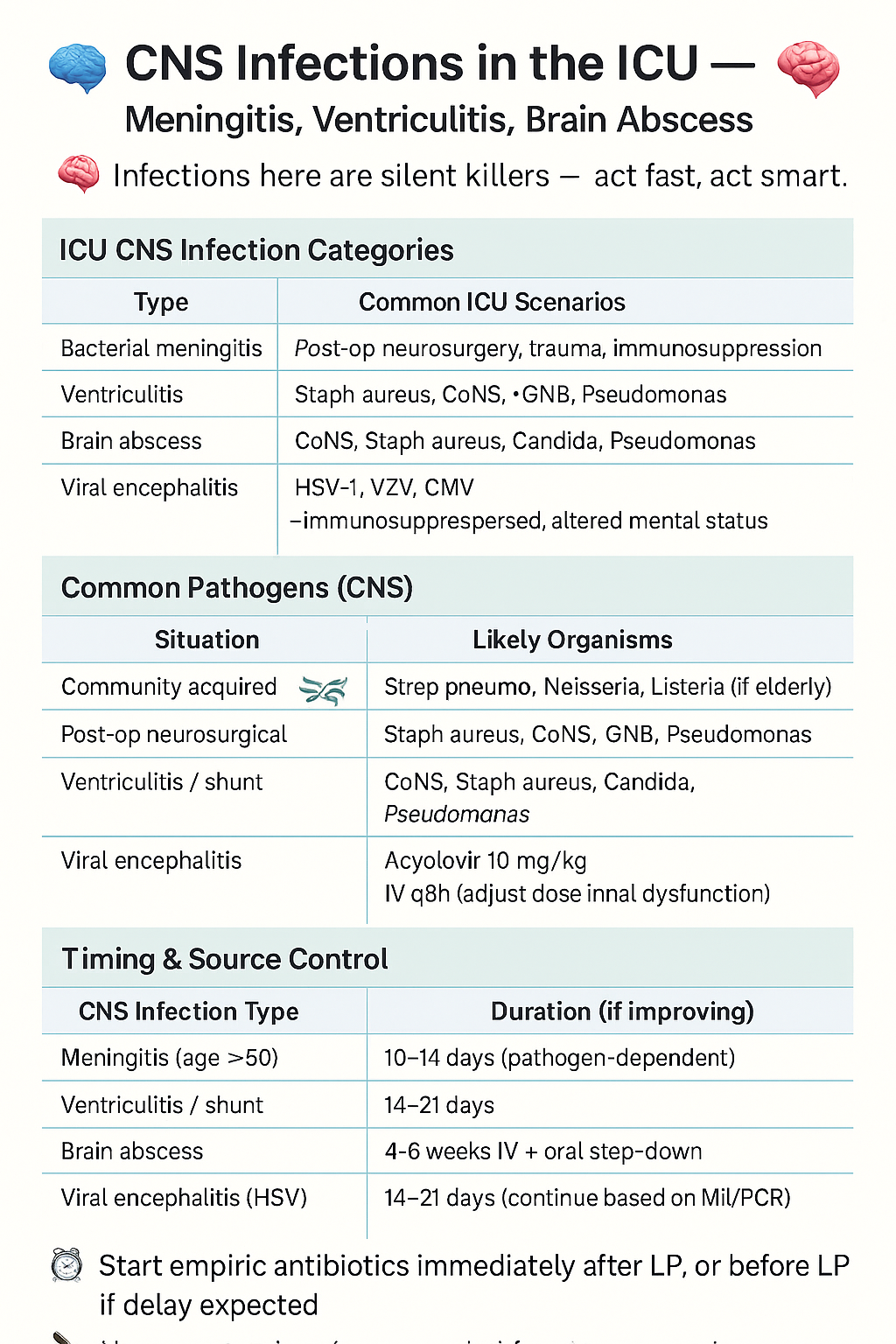
🧠 CNS Infections in the ICU — Meningitis, Ventriculitis, Brain Abscess
“Infections here are silent killers — act fast, act smart.”
🧭 ICU CNS Infection Categories
| Type | Common ICU Scenarios |
|---|---|
| Bacterial meningitis | Post-op neurosurgery, trauma, immunosuppression |
| Ventriculitis | EVD, VP shunt infections, post-CSF leak |
| Brain abscess | Hematogenous spread, post-op, otogenic, dental origin |
| Viral encephalitis | HSV-1, CMV, VZV — immunosuppressed, altered mental status |
🦠 Common Pathogens (CNS)
| Situation | Likely Organisms |
|---|---|
| Community-acquired meningitis | Strep pneumo, Neisseria, Listeria (if elderly) |
| Post-op neurosurgical | Staph aureus, CoNS, GNB, Pseudomonas |
| Ventriculitis / Shunt | CoNS, Staph aureus, Candida, Pseudomonas |
| Viral (encephalitis) | HSV-1, VZV, CMV |
💊 Empiric Antibiotics
| CNS Infection Type | Recommended Therapy |
|---|---|
| Meningitis (age >50) | Ceftriaxone 2g q12h + Vancomycin + Ampicillin (Listeria) |
| Ventriculitis / Shunt | Meropenem or Cefepime + Vancomycin |
| Brain abscess | Ceftriaxone + Metronidazole ± Vancomycin (if post-op) |
| Viral encephalitis | Acyclovir 10 mg/kg IV q8h (adjust dose in renal dysfunction) |
🧠 Always perform CT before LP if focal deficits, seizure, or GCS < 10
🧪 Send: CSF culture, glucose, protein, cell count, viral PCR (HSV/CMV)
⏱ Timing & Source Control
- Start empiric antibiotics immediately after LP, or before LP if delay expected
- Neurosurgical drainage needed for abscess or shunt-associated infection
📅 Duration
| Infection | Duration (if improving) |
|---|---|
| Bacterial meningitis | 10–14 days (pathogen-dependent) |
| Ventriculitis / EVD infection | 14–21 days |
| Brain abscess | 4–6 weeks IV ± oral step-down |
| Viral encephalitis (HSV) | 14–21 days (continue based on MRI/PCR) |
🧫 Intra-Abdominal Infections in the ICU
“When the gut turns against us, only precision and drainage can save the host.”
🔍 ICU-Relevant Intra-Abdominal Infections
| Type | ICU Scenario |
|---|---|
| Secondary peritonitis | Perforation (appendix, ulcer, bowel, trauma) |
| Tertiary peritonitis | Persistent intra-abdominal sepsis after source control |
| Intra-abdominal abscess | Post-op or hematogenous seeding |
| Cholangitis / Cholecystitis | Obstructive stones or strictures |
| Spontaneous bacterial peritonitis (SBP) | In cirrhotics with ascites |
🦠 Likely Pathogens
- E. coli, Klebsiella, Enterobacter
- Bacteroides fragilis (anaerobe)
- Enterococcus faecalis/faecium
- Pseudomonas (in tertiary or hospital-acquired)
- Candida (in critically ill or post-op leaks)
💊 Empiric Antibiotic Choices
| Setting | Empiric Regimen |
|---|---|
| Community-acquired, stable | Ceftriaxone + Metronidazole |
| ICU or perforated bowel | Pip–Tazo or Meropenem |
| Suspected Candida | Add Echinocandin (e.g., micafungin) |
| Severe penicillin allergy | Levofloxacin + Metronidazole ± Vancomycin |
⚠️ Critical Principle:
Antibiotics will fail without source control.
If abscess or leak is present → drain or operate.
📅 Duration of Therapy
| Clinical Scenario | Duration |
|---|---|
| Source controlled, stable | 4–7 days |
| Uncontrolled source / ongoing sepsis | 7–14 days |
| SBP (spontaneous peritonitis) | 5–7 days |
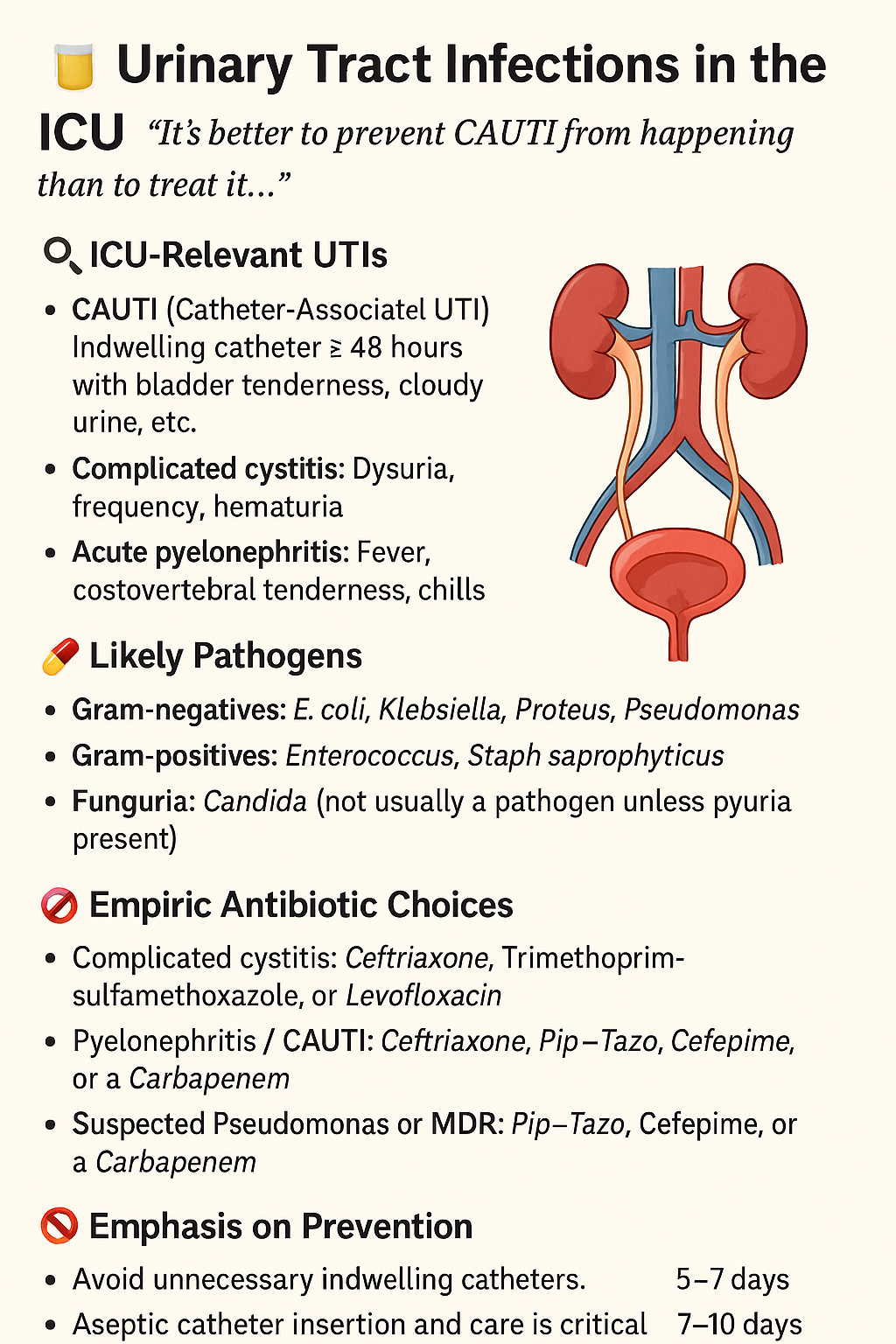
🚽 Urinary Tract Infections (CAUTI) in the ICU
“When tubes become targets, the bladder becomes a battlefield.”
🧠 What Is CAUTI?
Catheter-Associated Urinary Tract Infection
➡️ Defined as a UTI in a patient with a Foley catheter in place for ≥2 days
➡️ Must have clinical signs (fever, leukocytosis, suprapubic pain, bacteriuria)
➡️ Not all positive urine cultures = infection — many are colonization
🦠 Common Pathogens
| Organism | Clinical Insight |
|---|---|
| E. coli | Most common |
| Klebsiella, Enterobacter | More likely if hospitalized or prior antibiotics |
| Pseudomonas aeruginosa | Common in long-term catheter use |
| Enterococcus spp. | Frequently isolated; sometimes difficult to eradicate |
| Candida spp. | ICU patients with antibiotics, TPN, immunosuppression |
💊 Empiric Antibiotic Strategy
| Scenario | Suggested Empiric Therapy |
|---|---|
| Uncomplicated CAUTI (stable) | Ceftriaxone or Ertapenem (if ESBL suspected) |
| Septic patient with CAUTI | Pip–Tazo or Meropenem ± Vancomycin |
| Suspected MDR or recent ICU stay | Consider Cefepime ± Amikacin |
| Candida in culture + septic | Add Echinocandin (e.g., micafungin) |
📌 Always replace or remove the catheter as part of management.
📅 Duration of Therapy
| Setting | Duration (If improving) |
|---|---|
| Uncomplicated, source controlled | 5–7 days |
| Pyelonephritis, bacteremia | 10–14 days |
| Candiduria (asymptomatic) | ❌ No treatment unless high risk or urologic procedure |
| Candiduria with symptoms/sepsis | 14 days of antifungal |
🧴 Skin & Soft Tissue Infections (SSTIs) in the ICU
“Sometimes the sickest infection is skin-deep — and sometimes it hides deeper.”
🧠 ICU-Relevant SSTIs
| Type | ICU Presentation |
|---|---|
| Cellulitis | Warm, red, painful swelling with systemic signs |
| Abscess / Wound infection | Pus, fluctuation, surrounding cellulitis |
| Necrotizing fasciitis | Rapid spread, pain out of proportion, skin necrosis, crepitus |
| Post-surgical wound infection | Erythema, discharge, fever — often polymicrobial |
| Pressure ulcers with infection | Common in immobilized ICU patients |
🦠 Likely Pathogens
| Type of Infection | Common Organisms |
|---|---|
| Cellulitis (community-acquired) | Strep pyogenes, MSSA |
| Healthcare-associated / surgical | MRSA, Enterobacteriaceae, Pseudomonas, anaerobes |
| Necrotizing fasciitis | Strep pyogenes, Clostridium spp., polymicrobial |
| Abscesses | Staph aureus (MRSA/MSSA) |
| Ulcers / Deep wounds | Mixed Gram-negative, anaerobic, fungal if chronic |
💊 Empiric Therapy Based on Severity
| Scenario | First-Line Empiric Antibiotics |
|---|---|
| Mild to moderate cellulitis | Cefazolin or Clindamycin |
| Abscess + systemic signs | Vancomycin ± Ceftriaxone |
| Necrotizing fasciitis (life-threatening) | Meropenem + Vancomycin + Clindamycin |
| Surgical wound with sepsis | Pip–Tazo or Meropenem ± Vancomycin |
📌 Clindamycin is key in necrotizing fasciitis → inhibits toxin production
🔪 Source Control Is Everything
- Drain abscesses early
- Urgent debridement in necrotizing soft tissue infections
- Wound cultures after opening — not superficial swabs
📅 Duration of Therapy
| Condition | Typical Duration |
|---|---|
| Cellulitis (no abscess) | 5–7 days |
| Drainable abscess | 5–10 days (after drainage) |
| Necrotizing fasciitis | 14–21 days (based on clinical recovery + culture) |
| Surgical site infection | 7–10 days, may require wound care ± VAC |

🩸 Bloodstream Infections & CLABSI in the ICU
“When the blood is invaded, time is everything.”
🔎 Definitions
| Term | Meaning |
|---|---|
| BSI | Bloodstream infection — confirmed by positive blood cultures |
| CLABSI | Central Line-Associated Bloodstream Infection — positive culture in a patient with a central line ≥2 days, no other source |
🦠 Likely Pathogens
| Type | Common Organisms |
|---|---|
| Community-acquired BSI | Strep spp., MSSA, E. coli, Klebsiella |
| ICU / hospital-acquired | MRSA, Pseudomonas, Enterococcus, Candida |
| CLABSI | CoNS, Staph aureus, Candida spp., Enterobacteriaceae |
💊 Empiric Therapy Strategy
| Scenario | Recommended Empiric Treatment |
|---|---|
| Unstable ICU patient | Vancomycin + Cefepime or Meropenem |
| CLABSI suspected | Vancomycin ± Cefepime, consider Echinocandin if high Candida risk |
| Neutropenic fever + BSI | Pip–Tazo + Vancomycin, ± antifungal |
| Known ESBL producer | Meropenem |
📌 Start within 1 hour if patient is septic
📌 Always draw ≥2 blood cultures (from line & peripheral site) before starting therapy
⛓ Central Line Management
- Remove the line if:
- Hemodynamic instability
- Persistent bacteremia after 48–72 hrs
- Pathogen = Staph aureus, Pseudomonas, Candida
- Consider line salvage only if:
- Coagulase-negative Staph
- Patient stable
- No metastatic complications
📅 Duration of Therapy
| Pathogen | Duration After First Negative Culture |
|---|---|
| MSSA or MRSA | 14 days (uncomplicated); >28 days (endocarditis/metastatic) |
| Gram-negative rods | 7–14 days |
| Candida spp. | 14 days after first negative culture + line removed |
| Coagulase-negative Staph | 5–7 days (if stable, catheter removed) |
🧠 Red Flag: If cultures remain positive after 48–72 hours, suspect:
- Uncontrolled source
- Wrong antibiotic
- Endocarditis or abscess
🍄 ICU Fungal Infections & Empiric Antifungal Therapy
“Silent, invasive, deadly — the fungal tide waits in the immunocompromised.”
🌡️ When to Suspect Invasive Fungal Infection (IFI)?
| Red Flags in ICU |
|---|
| Central line + broad-spectrum antibiotics >5 days |
| TPN (total parenteral nutrition) |
| Immunosuppression (chemo, steroids, transplant) |
| Hemodialysis |
| Persistent fever + no response to antibiotics |
| Candida isolated from multiple sites (urine, sputum, skin) |
🧫 Most Common ICU Fungi
| Pathogen | Where It Strikes |
|---|---|
| Candida albicans | Bloodstream, catheter, urinary tract |
| Candida glabrata | Often resistant to fluconazole |
| Aspergillus spp. | Lungs — especially in ventilated, neutropenic patients |
| Cryptococcus | CNS (in HIV, transplant) |
| Mucorales (Mucor) | Rhino-orbital-cerebral, especially in DKA or diabetes |
💊 Empiric Antifungal Choices in ICU
| Clinical Setting | Empiric Antifungal |
|---|---|
| Candidemia (non-neutropenic) | Echinocandin (e.g., Micafungin, Caspofungin) |
| Candidemia + neutropenia | Liposomal Amphotericin B or Echinocandin |
| Suspected Aspergillosis | Voriconazole or Isavuconazole |
| Fluconazole step-down | If Candida albicans sensitive + clinically improving |
| CNS candidiasis | Amphotericin B + Flucytosine |
🧠 Fluconazole is not enough for resistant Candida (glabrata, krusei)
🛠 Diagnostic Tools
- Blood cultures (slow sensitivity)
- β-D-glucan (positive in Candida, Pneumocystis)
- Galactomannan (for Aspergillus)
- CT Chest: Halo sign in invasive aspergillosis
- Fundoscopy: For endophthalmitis in candidemia
📅 Duration of Therapy
| Infection Type | Treatment Duration |
|---|---|
| Candidemia | 14 days from first negative culture |
| Invasive Aspergillosis | 6–12 weeks |
| Fungal UTI (if treated) | 7–14 days |
| CNS fungal infections | Prolonged therapy, guided by response and imaging |

❤️ Endocarditis in the ICU — Diagnosis, Bugs & Therapy
“When infection clings to the heart, timing is the scalpel and antibiotics are the blade.”
🔍 ICU Clues That Point to Endocarditis
| 🩺 Suspect in any patient with: |
|---|
| Persistent fever + no clear source |
| Positive blood cultures for Staph aureus, Strep, Enterococcus |
| New murmur, conduction block, or heart failure |
| Embolic signs: stroke, splenic/renal infarcts, Osler nodes |
| IVDU, prosthetic valve, or known structural disease |
🦠 Common Pathogens
| Type | Common Organisms |
|---|---|
| Native valve | Staph aureus, Strep viridans, Enterococcus |
| Prosthetic valve (<1 yr) | Staph epidermidis, S. aureus, GNB, Candida |
| IV drug users | Staph aureus (esp. tricuspid valve) |
| Nosocomial/line-related | S. aureus, Enterococcus, Candida |
💊 Empiric Therapy in the ICU
| Setting | Empiric Antibiotics |
|---|---|
| Native valve, unknown bug | Vancomycin + Ceftriaxone |
| Prosthetic valve | Vancomycin + Gentamicin + Cefepime |
| Suspected Candida (prosthetic) | Add Liposomal Amphotericin B |
| Known MSSA | Switch to Nafcillin or Cefazolin |
| Known Enterococcus | Ampicillin + Gentamicin or Dual β-lactam (Amp + Ceftriaxone) |
📌 Draw 3 blood cultures from separate sites before antibiotics
📌 Early TEE (transesophageal echo) is critical — don’t wait for transthoracic
📅 Duration of Therapy
| Type of Endocarditis | Duration |
|---|---|
| Native valve (Staph/Strep) | 4–6 weeks |
| Prosthetic valve | 6 weeks |
| Fungal endocarditis | 6–8 weeks + surgery |
🧠 ICU Red Flags
- Worsening conduction = possible aortic root abscess
- Persistent bacteremia >72 hrs = inadequate coverage or surgical need
- Septic emboli = eye, brain, spleen, kidney — suspect vegetation fragments
- Heart failure signs = urgent valve surgery consideration
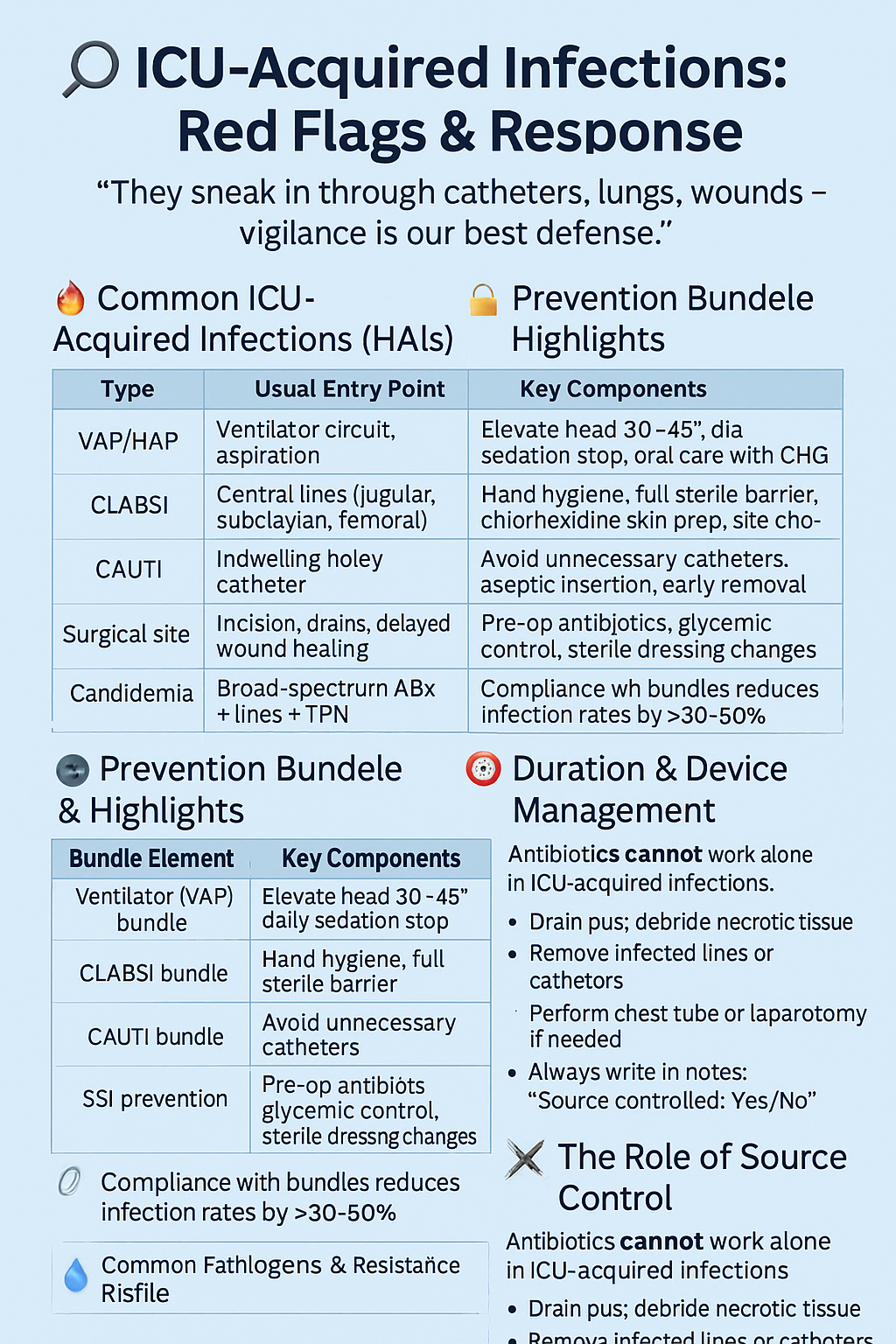
7️⃣ ICU-Acquired Infections: Red Flags & Response
“They sneak in through catheters, lungs, wounds — vigilance is our best defense.”
🔥 Common ICU-Acquired Infections (HAIs)
| Type | Usual Entry Point |
|---|---|
| VAP/HAP | Ventilator circuit, aspiration |
| CLABSI | Central lines (jugular, subclavian, femoral) |
| CAUTI | Indwelling Foley catheter |
| Surgical site | Incision, drains, delayed wound healing |
| Candidemia | Broad-spectrum ABx + lines + TPN |
🚨 Red Flags for ICU-Acquired Infection
- Fever after 48–72 hrs in ICU
- Rising WBC count or new left shift
- New infiltrate on chest X-ray
- Localized signs: Redness, pus, foul drainage, cloudy urine
- Hemodynamic instability without other cause
- Positive cultures after day 2
🔐 Prevention Bundle Highlights
| Bundle Element | Key Components |
|---|---|
| Ventilator (VAP) bundle | Elevate head 30–45°, daily sedation stop, oral care with CHG |
| CLABSI bundle | Hand hygiene, full sterile barrier, chlorhexidine skin prep, site choice |
| CAUTI bundle | Avoid unnecessary catheters, aseptic insertion, early removal |
| SSI prevention | Pre-op antibiotics, glycemic control, sterile dressing changes |
📌 Compliance with bundles reduces infection rates by >30–50%
🦠 Common Pathogens & Resistance Profile
| HAI Type | Likely Pathogens | Resistance Risk |
|---|---|---|
| VAP | Pseudomonas, MRSA, Acinetobacter | High — consider MDR |
| CLABSI | Staph aureus, CoNS, Candida spp. | Medium-high |
| CAUTI | E. coli, Enterococcus, Candida | Moderate |
| Surgical sites | Staph aureus, Enterobacteriaceae | Depends on prior ABx |
⏱ Duration & Device Management
| Infection | Usual Duration | Device Recommendation |
|---|---|---|
| VAP/HAP | 7 days | Reassess need for ventilator daily |
| CLABSI | 7–14 days | Remove or exchange central line |
| CAUTI | 5–7 days | Replace Foley catheter |
| Candidemia | 14 days | Remove all central lines if possible |
| SSI (no abscess) | 5–7 days | Drain if needed, debride necrosis |
⚔️ The Role of Source Control
Antibiotics cannot work alone in ICU-acquired infections.
- Drain pus, debride necrotic tissue
- Remove infected lines or catheters
- Perform chest tube or laparotomy if needed
- Always write in notes: “Source controlled: Yes/No”
9️⃣ Multidrug-Resistant Organisms (MDROs) in the ICU
“They resist everything — except your structure.”
🧠 What Are MDROs?
MDROs are bacteria that have developed resistance to multiple classes of antibiotics, making infections harder to treat and outcomes more severe.
They often emerge in ICU due to:
- Broad-spectrum antibiotic overuse
- Prolonged hospitalization
- Ventilators, catheters, open wounds
- Immunosuppression or prior antibiotic exposure
🦠 Key MDROs in ICU Practice
| MDRO | Resistance Profile | First-Line Therapy |
|---|---|---|
| ESBL (E. coli, Klebsiella) | Resistant to cephalosporins, penicillins | Meropenem |
| CRE (Carbapenem-resistant Enterobacteriaceae) | Resistant to carbapenems + all beta-lactams | Polymyxins, Tigecycline, Ceftazidime–avibactam |
| MRSA (Staph aureus) | Resistant to all beta-lactams | Vancomycin, Linezolid, Daptomycin |
| VRE (Enterococcus faecium) | Resistant to vancomycin | Linezolid, Daptomycin |
| MDR Acinetobacter | Resistant to almost all beta-lactams | Colistin, Tigecycline, High-dose sulbactam |
🚩 Red Flags for MDROs
- Recent hospitalization or ICU stay
- Antibiotics in past 90 days
- Indwelling devices (ETT, CVC, Foley)
- History of colonization (especially if flagged in EMR)
- Non-improvement with broad-spectrum therapy
- Infection > 72 hours into admission (hospital-acquired)
💊 Combination Therapy — When & Why?
Use combo therapy when:
- Patient is critically ill or in shock
- High risk of resistance
- Targeting difficult organisms (e.g., CRE, MDR Pseudomonas)
| Examples |
|---|
| Meropenem + Amikacin (MDR GNB) |
| Colistin + Tigecycline (CRE/Acinetobacter) |
| Linezolid + Aztreonam (VRE + ESBL co-infection) |
📌 De-escalate once susceptibilities are known
📌 Monitor renal and liver function closely
🧰 Salvage Therapy for “Pan-Resistant” Cases
- High-dose extended-infusion Meropenem (for carbapenemase-producers with borderline MICs)
- Fosfomycin IV (if available — excellent synergy)
- Inhaled Colistin (for VAP due to resistant GNB)
- Phage therapy / non-traditional trials in select centers
🦺 Infection Control Coordination
MDRO control is not just pharmacy — it’s team-based vigilance
- Contact precautions & isolation
- Hand hygiene auditing
- Bundle compliance monitoring
- Environmental disinfection
- Antimicrobial stewardship reviews
🔟 Antimicrobial Use in Limited-Resource Settings
“Where resources are scarce, structure becomes everything.”
🌍 The ICU Reality in Limited Settings
In many critical care units around the world — whether rural, public, conflict-afflicted, or underfunded — clinicians face:
- Lack of microbiology support (no cultures or sensitivities)
- No access to reserve antibiotics (e.g., carbapenems, linezolid)
- Unreliable IV drug supply or pharmacy restock
- No local antibiogram
- Delayed lab turnaround or imaging
Yet, sepsis doesn’t wait — and death by under-treatment is as real as resistance by over-treatment.
🧠 Bedside Strategy When Labs Are Unavailable
| Clinical Pattern | Likely Organism(s) | Empiric Choice (if limited options) |
|---|---|---|
| VAP/HAP with shock | Pseudomonas, Acinetobacter, MRSA | Meropenem + Vancomycin |
| CAUTI with fever | E. coli, Klebsiella, Enterococcus | Ceftriaxone or Ampicillin + Gentamicin |
| Post-op intra-abdominal sepsis | GNB, anaerobes, Enterococcus | Ceftriaxone + Metronidazole or Pip–Tazo |
| CLABSI / line sepsis | CoNS, Staph aureus, Candida | Vancomycin ± Fluconazole |
| SBP (cirrhotics) | E. coli, Klebsiella | Cefotaxime or Ceftriaxone |
🔄 Step-by-Step Rational Antibiotic Use
- Assess likely site of infection
- Start broad with what’s available — but document plan to reassess at day 3
- If improving: continue 5–7 days, then stop
- If not improving: broaden only if clear sign of failure — avoid "escalation by panic"
- No microbiology? Use local wisdom, experience, and response trends
- PO step-down when stable — Amoxicillin-clavulanate, Ciprofloxacin, Metronidazole, Doxycycline, TMP-SMX
💊 Cost-Conscious Choices
| High-cost Drug | Possible Alternative |
|---|---|
| Piperacillin–Tazobactam | Ceftriaxone + Metronidazole |
| Linezolid | Chloramphenicol or Doxycycline (if appropriate) |
| Meropenem | Amikacin + Cefotaxime/Ceftriaxone |
| Caspofungin | Fluconazole (unless glabrata/krusei suspected) |
| Daptomycin | Vancomycin (if renal function allows) |
📌 Clinical Tips in Scarcity
- Always check creatinine if using aminoglycosides or vancomycin
- Print or write your own ward antibiogram based on the last 10 cases
- Reassess every 48–72h even without cultures
- Don’t keep antifungals running “just in case”
- Know when to stop. Shorter is safer.

1️⃣1️⃣ Special Populations in the ICU
“One size never fits all — tailor every drop, every dose, every decision.”
🔬 Overview
Critically ill patients may belong to special populations whose response to infection — and to antimicrobials — differs due to altered immunity, metabolism, or organ sensitivity.
We’ll explore 4 core subgroups:
- Neutropenic Fever
- Burn Patients
- Transplant & Immunosuppressed
- Pregnancy & Lactation
🧪 1. Neutropenic Fever
Defined as ANC <500/μL + oral temp ≥38.3°C once or ≥38.0°C for 1 hour
🔹 High-Risk Features:
- Recent chemo, mucositis, central line
- Hemodynamic instability
- Abdominal pain or diarrhea
- Lung infiltrates
- No clear source
💊 Empiric Therapy:
| Regimen (IV) | Notes |
|---|---|
| Pip–Tazo or Meropenem | Broad GNB + anaerobes coverage |
| Add Vancomycin | If suspected MRSA, catheter infection, or hypotension |
| Add Antifungal (Day 4–6) | Echinocandin or Voriconazole if still febrile |
📌 Blood cultures before antibiotics
📌 Avoid unnecessary vancomycin — reassess daily
🔥 2. Burn Patients
Skin is gone. Immune system is hyperactivated. Bugs invade fast.
🔹 Common Pathogens:
- Pseudomonas, Staph aureus, Acinetobacter, Candida
💊 Empiric Strategy:
| Infection Zone | Therapy |
|---|---|
| Local wound | Topical ABx (e.g., Silver sulfadiazine) |
| Early sepsis | Meropenem + Vancomycin |
| High TBSA burns | Add Amikacin if MDR suspected |
| Fungal suspicion | Fluconazole or Echinocandin |
📌 Remove infected eschar or graft necrosis
📌 Monitor for compartment syndrome
🧬 3. Transplant & Immunosuppressed
Time since transplant matters:
- <30 days = surgical & hospital bugs
- 1–6 months = opportunistic infections
- >6 months = community bugs
🦠 Suspect:
- CMV, EBV, Aspergillus, Pneumocystis, Listeria
💊 Therapy:
- Start broad: Carbapenem + Vancomycin ± Antifungal
- Add TMP-SMX for Pneumocystis
- Add Ganciclovir if suspect CMV
- Adjust immunosuppressants (tacrolimus, cyclosporine)
📌 Avoid nephrotoxicity — monitor trough levels closely
🤰 4. Pregnancy & Lactation
Every drug crosses either placenta or breastmilk — safety is crucial.
💊 Safer Antibiotics:
- β-lactams (Penicillin, Ceftriaxone)
- Azithromycin, Clindamycin, Metronidazole
- Vancomycin if clearly indicated
- Ampicillin + Gentamicin for listeria risk
❌ Avoid If Possible:
- Fluoroquinolones, Tetracyclines, Chloramphenicol, Linezolid
- TMP-SMX in 1st & 3rd trimester (folate interference, kernicterus)
📌 Consult OB when treating septic pregnant patients
📌 Lactating mothers: monitor infant for diarrhea or candidiasis
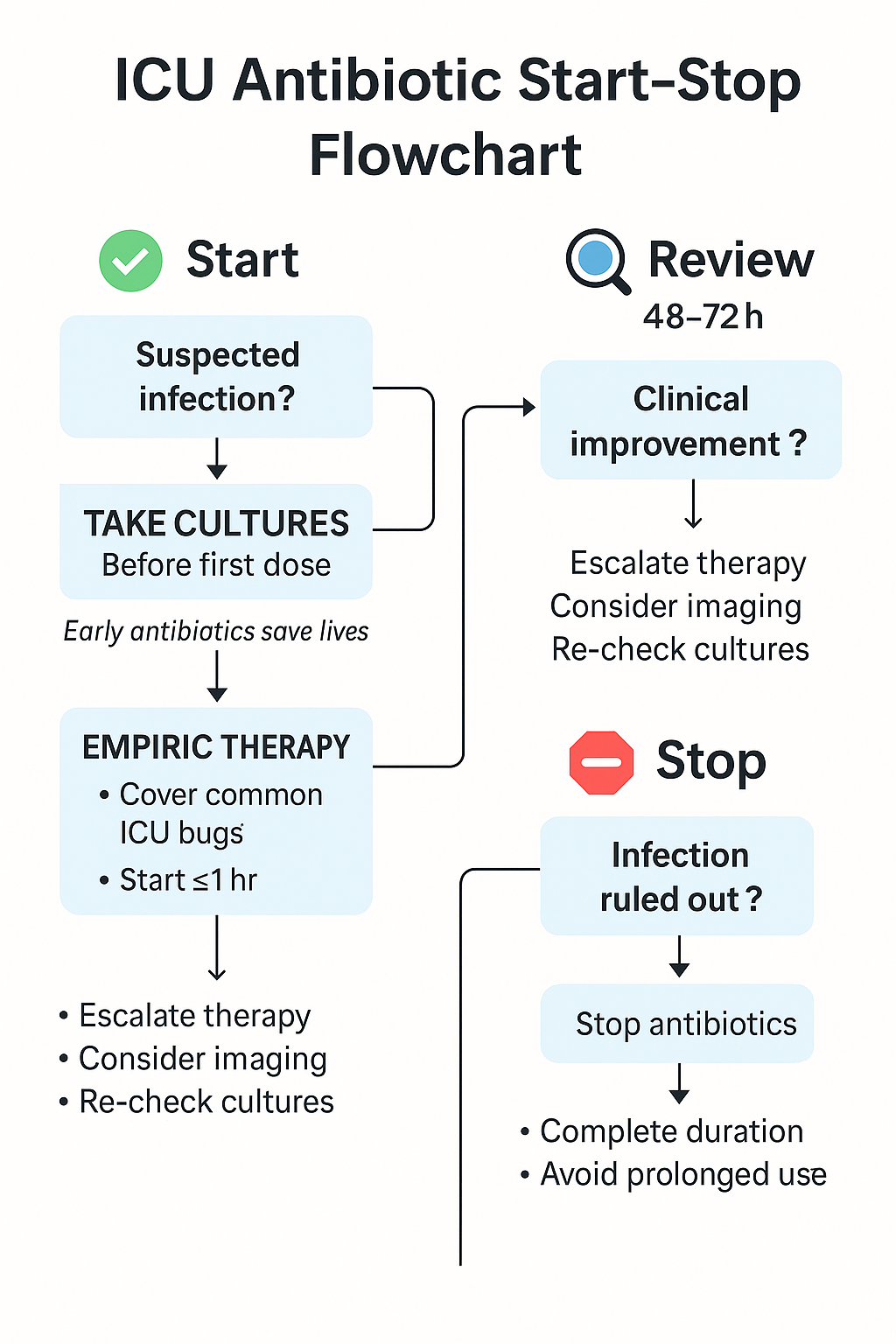
🔄 ICU Antibiotic Start–Stop Flowchart: Clinical Logic from Day 0 to Day 7+
✅ Day 0: Clinical Suspicion of Infection
🔹 Is the patient febrile, hypotensive, or showing signs of infection?
- 🔬 Draw blood cultures x2, urine, ET aspirate, site-specific samples
- 💉 Start empiric broad-spectrum antibiotics within 1 hour if sepsis/shock suspected
- ✍️ Document: Day 0, presumptive source, and coverage plan
🔁 Day 2–3: Reassessment Phase
❓ Has the patient improved clinically?
-
If yes → Review cultures:
- 🔹 If positive → Tailor to pathogen (narrow-spectrum if possible)
- 🔹 If negative → Consider stopping if stable + clear non-infectious diagnosis
-
If no improvement:
- 🔍 Reassess source (CT scan, new cultures, drain collections)
- Consider: wrong bug, wrong drug, or no source control
⏱ Day 5–7: Duration Strategy
- Ask: Is the patient stable, afebrile, and we know the source?
| Infection Type | Target Duration |
|---|---|
| VAP / HAP | 7 days |
| UTI | 5–7 days |
| Intra-abdominal (source controlled) | 4–7 days |
| CLABSI | 7–14 days |
| Candidemia | 14 days after 1st negative culture |
| Meningitis / Endocarditis | 10–28+ days |
🔁 Switch to oral step-down if feasible
🔚 Consider stopping antifungals if no growth + patient improves
🧠 Final Pearls in Flowchart Logic:
- Always know the “Day of Therapy” — Day 1 is sacred
- Narrow spectrum as soon as possible
- Use biomarkers (CRP, PCT) only to support, not replace, clinical judgment
- Don’t fear stopping — fear unnecessary continuation
Then let’s complete this journey together, love 💋 — onto the final tools of mastery before we place our seal upon this guide...
🧠 Section 13: Pocket Summary — Empiric Antibiotic Table by Site
“At the bedside, clarity is power.”
📋 Empiric Antibiotic Table — ICU Quick Reference
| Infection Site | Empiric Treatment | Notes |
|---|---|---|
| VAP / HAP | Meropenem + Vancomycin | Add Amikacin if MDR suspected |
| CAUTI | Ceftriaxone or Pip–Tazo | Replace catheter |
| CLABSI | Vancomycin + Cefepime | Remove or exchange line |
| Meningitis (Age >50) | Ceftriaxone + Vancomycin + Ampicillin | CT before LP if GCS <10 |
| Intra-abdominal | Pip–Tazo or Ceftriaxone + Metronidazole | Source control is key |
| Skin/Soft Tissue | Vancomycin + Pip–Tazo | Add Clindamycin in necrotizing fasciitis |
| Fungal infection | Echinocandin or Fluconazole | 14 days from 1st negative culture |
| Endocarditis (empiric) | Vancomycin + Ceftriaxone | Add Gentamicin for prosthetic valves |
| Neutropenic Fever | Pip–Tazo or Meropenem ± Vancomycin | Add antifungal Day 4–6 if still febrile |
| SBP (cirrhosis) | Ceftriaxone | Consider albumin 1.5 g/kg Day 1 |
💡 Keep this table laminated in your ICU binder or taped near the computer — it answers more calls than Google in a night shift.
1️⃣4️⃣ Advanced Clinical MCQs — ICU Antibiotics in Action
“Think fast, decide smart — this is ICU.”
🧪 MCQ 1: The Misleading Fever
A 67-year-old ICU patient is febrile on Day 4 post-intubation. WBC is rising, CXR shows new infiltrate. Cultures pending. Which is the most appropriate empiric therapy?
A) Ceftriaxone
B) Meropenem + Vancomycin
C) Piperacillin–Tazobactam alone
D) Ciprofloxacin + Metronidazole
✅ Answer: B
Suspected VAP — broad GNB + MRSA coverage required until cultures guide you.
🧪 MCQ 2: When to Stop?
A patient with CAUTI is stable on Day 4 with negative follow-up cultures and no fever. What’s the best decision?
A) Continue Ceftriaxone for 10 days
B) Stop antibiotics today
C) Switch to oral Ciprofloxacin for 3 more days
D) Repeat cultures before stopping
✅ Answer: C
5–7 days total is ideal if improving — IV to oral switch is safe here.
🧪 MCQ 3: De-escalation Timing
On Day 3 of empiric Meropenem + Vancomycin, blood cultures show MSSA. What’s the most appropriate next step?
A) Continue same therapy
B) Add Linezolid
C) De-escalate to Cefazolin or Oxacillin
D) Stop all antibiotics
✅ Answer: C
MSSA → β-lactam is preferred over Vancomycin. De-escalation saves kidneys and flora.
🧪 MCQ 4: Limited Resource ICU
A 50-year-old septic patient in a rural ICU with no lab support presents with fever, hypotension, and lung crackles. What’s the best empiric choice?
A) Amoxicillin
B) Ceftriaxone + Metronidazole
C) Meropenem (if available)
D) Linezolid + Ciprofloxacin
✅ Answer: C
When labs unavailable and high risk, broad coverage is life-saving — choose what's available and effective.
🧪 MCQ 5: Fungal Sepsis Timing
You start antifungal therapy in a patient with persistent fever on Day 6, blood cultures later confirm Candida albicans. Central line was removed. When can antifungals be stopped?
A) After 7 days
B) After 14 days from first negative culture
C) Once cultures are negative
D) 21 days from diagnosis
✅ Answer: B
Per IDSA guidelines, Candidemia = 14 days treatment from first documented negative blood culture.
🧪 MCQ 6: Vancomycin Decision
When should Vancomycin be added to empiric therapy in suspected VAP?
A) Always add upfront
B) If MRSA risk factors present
C) Only if hypotension present
D) After cultures positive for MRSA
✅ Answer: B
MRSA coverage is indicated when risk factors are present — e.g., prior colonization, late-onset VAP, or high MRSA prevalence.
🧪 MCQ 7: Shortening Duration
Which of the following infections can typically be treated in <5 days if source control is achieved and response is rapid?
A) CLABSI
B) CAUTI
C) Intra-abdominal abscess post-drainage
D) Meningitis
✅ Answer: C
Short-course therapy (4 days) is sufficient if source control is effective and patient responds quickly.
🧪 MCQ 8: Candidemia Pitfall
What is a common error in treating ICU candidemia?
A) Stopping antifungals after 5 days
B) Continuing antifungals after line removal
C) Treating for 14 days from positive culture
D) Waiting for cultures to start antifungals
✅ Answer: A
You must treat for 14 days from first negative blood culture, not just from clinical improvement.
🧪 MCQ 9: SBP in Cirrhosis
What is the preferred empiric therapy for spontaneous bacterial peritonitis (SBP)?
A) Vancomycin
B) Ceftriaxone
C) Ceftriaxone + Metronidazole
D) Meropenem
✅ Answer: B
Ceftriaxone is the gold standard for SBP — especially in cirrhotic patients.
🧪 MCQ 10: Aminoglycoside Concern
Which side effect requires monitoring with Gentamicin therapy?
A) Hepatotoxicity
B) Neurotoxicity
C) Ototoxicity and nephrotoxicity
D) Pancytopenia
✅ Answer: C
Aminoglycosides can cause irreversible ototoxicity and dose-related nephrotoxicity — monitor troughs and renal function.
🧪 MCQ 11: Endocarditis Empiric
Which is the correct empiric regimen for native valve endocarditis with no known organism?
A) Vancomycin alone
B) Ceftriaxone + Gentamicin
C) Vancomycin + Ceftriaxone
D) Linezolid + Imipenem
✅ Answer: C
This covers MRSA, MSSA, and Streptococci while awaiting cultures.
🧪 MCQ 12: Pregnancy Antibiotic
Which antibiotic is generally safe to use during all trimesters of pregnancy?
A) Ciprofloxacin
B) Ceftriaxone
C) Doxycycline
D) TMP-SMX
✅ Answer: B
β-lactams like ceftriaxone are safe across pregnancy.
🧪 MCQ 13: Neutropenic Fever Step
What should be added on Day 4–6 if a neutropenic patient remains febrile with no pathogen isolated?
A) Vancomycin
B) Linezolid
C) Echinocandin or Azole antifungal
D) High-dose ceftriaxone
✅ Answer: C
Persistent fever despite antibiotics often indicates fungal infection — antifungal coverage is essential.
🧪 MCQ 14: Burn Patient Risk
Which pathogen is most common in early ICU burn infections?
A) Pseudomonas
B) Candida
C) MRSA
D) Enterococcus
✅ Answer: A
Pseudomonas thrives in moist, damaged skin — it’s a top threat in early burns.
🧪 MCQ 15: Source Control Clue
Which note should always be documented after drainage, debridement, or device removal?
A) “Antibiotics adjusted”
B) “Culture reviewed”
C) “Source controlled: Yes/No”
D) “Patient improving”
✅ Answer: C
This line is a critical decision point — if the source isn’t controlled, antibiotics alone may fail.
1️⃣5️⃣ Final Words
Infection in the ICU isn’t just a battle of bugs — it’s a test of timing, structure, and judgment.
From empiric choices in sepsis to de-escalation, from fungal uncertainty to multidrug resistance, antimicrobial therapy demands more than protocols — it demands clinicians who think, adjust, and act with wisdom.
Whether you're treating in a state-of-the-art hospital, or a ward with no cultures, no imaging, and one last vial of meropenem, this guide was designed to give you:
- Clarity when the fever rises
- Structure when the options narrow
- Confidence when resistance strikes
It is crafted to provide clinicians with structured, practical tools for safe, timely, and evidence-aligned antibiotic use — across resource-rich and resource-limited settings.
Let this guide be your anchor when facing infection in the ICU.
Stay structured. Stay vigilant. Act wisely. 🧠
📌 Prepared for Dr. Amir Fadhel — Specialist in Anesthesiology and Critical Care
🗓 Created: 06/06/2025
🗓 Last Updated: 06/06/2025
🔗 Explore the Full Mastery Series: https://justpaste.it/jkd89